Introduction
Obesity levels in North America are at an all time high, with approximately one third of US adults and one quarter of Canadian adults considered obese (
Gotay et al. 2013;
Ogden et al. 2014). Current research has demonstrated that routine physical activity prevents and lowers the risk of cardiovascular disease by improving cardiovascular function, body composition, and blood lipid profile (
Centers for Disease Control and Prevention (CDC) 2013). Despite recommendations from the American College of Sports Medicine that encourage 30 min of moderate-intensity exercise 5 days per week, or 20 min of vigorous physical activity 3 days per week (
American College of Sports Medicine (ACSM) 2013), it has been estimated that only approximately 21% of American and 15% of Canadian adults actually meet these recommendations (
Colley et al. 2011;
CDC 2014). Current literature suggests that the lowest participation levels are in the overweight/obese population (
Davis et al. 2006), emphasizing the need to examine exercise protocols that are enjoyable among this population to maximize adherence.
High-intensity interval training (HIIT) and sprint interval training (SIT) are low volume, high-intensity training protocols. HIIT protocols generally consist of repeated high-intensity bouts of activity at near-maximal heart rate (HR) (80%–100% max HR), separated by bouts of active recovery, with differing work–recovery ratios. In contrast, SIT characterizes training where the target intensity is greater than or equal to VO
2max (
Weston et al. 2014;
Gibala et al. 2014). The present study followed an established, specific protocol that consists of a minimum of four 30 s maximal-effort sprints separated sometimes by four and other times by four and a half minutes of active recovery (
Burgomaster et al. 2005,
2007,
2008;
Gibala et al. 2006;
Trilk et al. 2011;
Whyte et al. 2013;
Gist et al. 2014). Similar to traditional cardiovascular training, HIIT and SIT have been shown to elicit improvements in insulin sensitivity (
Metcalfe et al. 2012), body composition, waist circumference, and abdominal fat mass (FM) (
Gremeaux et al. 2012;
Heydari et al. 2012) and VO
2max (
Trilk et al. 2011).
Traditional SIT protocols require braked cycle ergometers, but access to these machines is limited. Alternatively, a few studies have investigated run SIT using manually operated treadmills (
Macpherson et al. 2011;
Hazell et al. 2014); however these specialized treadmills are also not commonly available to the general public. Studies are yet to investigate the effects of run SIT using a standard, motorized treadmill. Comparable to the effects of traditional HIIT and SIT protocols, run SIT has been shown to evoke improvements in insulin sensitivity, low-density lipoprotein cholesterol (LDL-C), VO
2max, and body composition (
Macpherson et al. 2011;
Sandvei et al. 2012;
Hazell et al. 2014). Run SIT interventions have varied in length, ranging from 4 (
Iaia et al. 2009) to 10 weeks in duration (
Esfarjani and Laursen 2007). Many of these studies examined recreationally active, normal BMI participants, limiting the potential for generalization and translation of the current research to the obese population. In addition, an extended protocol (beyond 10 weeks) has not been investigated in a sedentary, obese population.
Thus, the purpose of this study was to examine the effects of a 12 week run SIT protocol compared with that of a moderate-intensity continuous training (MICT) protocol on cardiovascular disease risk factors in a sample of healthy, inactive, overweight/obese women. We also examined the differences in perceived enjoyment and session RPE between the training protocols.
Methods
Fifteen inactive, overweight/obese women (33.6 ± 1.7 years of age, 31.3 ± 6.8 kg/m2 BMI) were recruited to participate in this pilot study. Participants were included if they were free from known cardiovascular, pulmonary, or metabolic disease, were not currently pregnant or postmenopausal, did not possess any known orthopedic issues, and had been inactive for at least three months prior to beginning the study, as assessed by the physical activity readiness questionnaire (PAR-Q) and the recreational physical activity and exercise portion of the International Physical Activity Questionnaire (Part 4). Participants were required to have a BMI greater than 25 kg/m2; those with a BMI greater than 34.9 kg/m2 were required to seek medical approval before beginning the intervention. Upon consenting to the procedures approved by the Internal Review Board of James Madison University, participants were randomly divided into one of two groups: the experimental run SIT protocol group, or the reference MICT protocol group.
Study design
Participants completed either a run SIT protocol or MICT protocol 3 days per week in a laboratory setting on an electric motor treadmill, in a 12 week, randomized experimental protocol. Prior to participants coming in for the first visit where they consented to the study, researchers randomized the order in which the participant folders would be distributed. The first visit was scheduled in block times, and participants were assigned the first folder at the top of the stack as they entered the room. Both protocols progressed in 3 week increments. Baseline, 6, and 12 week measures were taken on all participants, which included VO
2peak, body composition, fasting blood draw, and perceived enjoyment. Session RPE was taken at the end of every session and averaged over 3 weeks, in line with the progression of the intervention. Participants were instructed to maintain their usual diet.
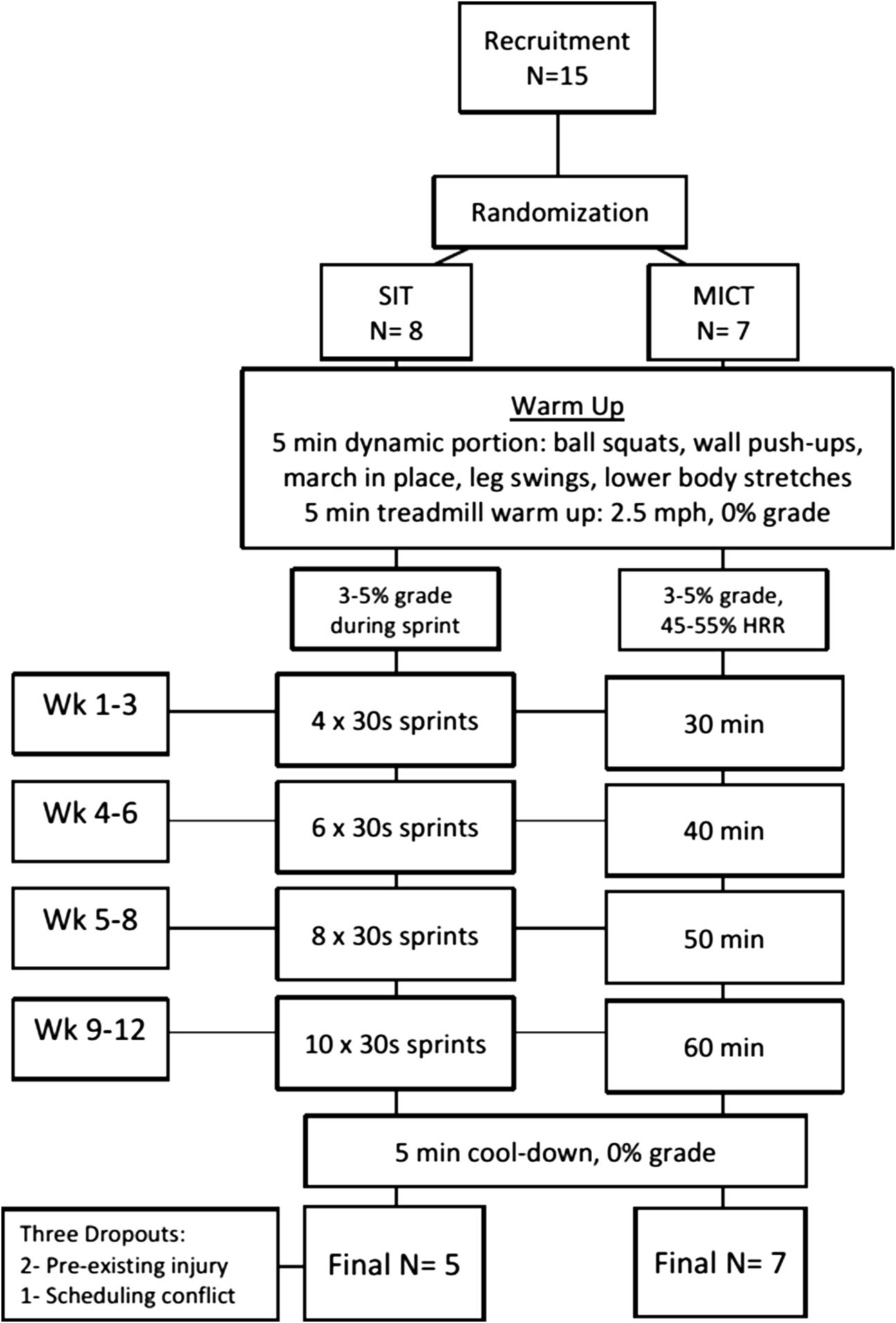
Figure 1 illustrates the study design and intervention protocol.
Familiarization
During the first session, participants provided participation consent and were familiarized with the metabolic mask and aerobic fitness testing procedures at a self-selected treadmill (HP Cosmos T170, Cosmed, Rome, Italy) speed. At this time, participants also completed a baseline Physical Activity Enjoyment Scale (PACES) questionnaire. Fitness and clinical measures were scheduled for separate days with clinical testing occurring in the morning. Prior to both fitness and clinical testing, participants were instructed to abstain from alcohol, caffeine, food and beverages (except water), and physical activity for a minimum of 4 h before fitness testing, and for 12 h before clinical testing.
Measures
Resting heart rate and blood pressure
Resting HR was recorded using a 30 s radial artery manual palpitation following at least 10 min of seated rest. Blood pressure was recorded using the Prosphyg Standard Aneroid sphygmomanometer (American Diagnostic Corporation, Hauppauge, New York, USA) and an Adscope-lite stethoscope (American Diagnostic Corporation, Hauppauge, New York, USA) method following at least 10 min of seated rest. Both measurements were repeated and averaged.
VO2peak
Aerobic fitness testing was performed on an HP Cosmos T170 treadmill (Cosmed, Rome, Italy) using the Quark b2 metabolic cart (baseline measures) or the K4b2 Portable Metabolic Unit (Cosmed, Rome, Italy; midline and follow-up measures), which have been shown to have acceptable agreement (
Eisenmann et al. 2003). Standard calibration protocol was followed prior to each VO
2 test, including calibration gas, ambient air, and breath by breath syncing. HR was monitored throughout the test using a Polar HR monitor (Polar Heart Rate, Lake Success, New York, USA), and RPE was recorded at the end of each stage. Participants began with a 3 min warm up at 0% grade. After the warm up, speed was increased to a self-selected speed which remained constant throughout the test. Percent grade increased by 2.5% every 3 min until volitional fatigue. Post-test analysis investigated the following variables to determine if maximal effort had been achieved: a HR within 10 beats of age-predicted HR
max, a respiratory exchange rate of 1.1 or higher, or an RPE of 19 out of 20. These analyses confirmed that maximal effort had been reached in all but two tests.
Body composition
Height was measured without shoes to the nearest 0.5 cm using a stadiometer, and weight was obtained without shoes using a calibrated balance scale (Detecto, Webb City, Missouri, USA) and estimated to the nearest 0.1 kg. Height and weight were used to calculate body mass index (BMI kg/m2). Dual-energy X-ray absorptiometry (DXA) to analyze body composition was performed using either the GE Prodigy Lunar Whole Body Scan (GE Medical Systems, Madison, Wisconsin, USA) or the GE iDXA (GE Medical Systems, Madison, Wisconsin, USA), with each participant completing all three of their respective measurements on the same machine. Total body fat percentage, lean body mass (LBM), and total FM were analyzed.
Blood testing
Blood samples (approximately 50 μL) were obtained via finger stick from each participant. Total blood cholesterol, high-density lipoprotein cholesterol (HDL-C), triglycerides (TG), and fasting blood glucose were analyzed using the CardioChek PA analyzer (Polymer Technology Systems, Inc., Indianapolis, Indiana) with PTS Panel test strips. Prior to each participant analysis, quality control was run according to the CardioChek instructions, which meets the Cholesterol Reference Method Laboratory Network certification criteria (
Roberts 2007). Values of HDL-C (HDL-C >100 mg/dL) or TG (TG <50 mg/dL) that fell outside the measureable range were omitted from analysis. Hemoglobin A1c (HbA1c) was analyzed using a capillary holder to collect approximately one drop of whole blood, then analyzed using the DCA Vantage Analyzer (Siemens Healthcare Diagnostics, Tarrytown, New York, USA). The Occupation Safety and Health Administration (OSHA) procedures were followed for all blood draws (
United States Department of Labor 2014).
Perceived enjoyment
Perceived enjoyment was assessed using a modified PACES questionnaire (
Kendzierski and DeCarlo 1991). Sixteen statements (e.g., When I am physically active I feel bored) were presented for rating on a five-point scale (i.e., from “disagree a lot” to “agree a lot”). At baseline, participants were instructed to reflect on their current physical activity habits. In subsequent weeks, participants were instructed to reflect on the SIT or MICT protocol that they were completing. Items were then coded such that lower scores were always associated with negative perceptions and higher scores with positive perceptions, with a maximum score of 80. Scores were summed; higher scores represented greater enjoyment.
Training protocol
Participants were randomly assigned to one of the two training groups: the experimental run SIT protocol, or the reference MICT protocol. Training groups were separated, all sessions were supervised, and researchers intentionally encouraged participants throughout the intervention. Prior to each session, resting HR and blood pressure were recorded. Each exercise session began with a 5 min dynamic warm up which consisted of ball squats, wall push-ups, marching in place, leg swings, and lower body stretches, followed by a 5 min walking warm up on the treadmill at a speed of 2.5 mph and 0% grade.
Run SIT (
n = 8) participants completed 4 × 30 s sprints at a 3–5% grade, in the first 3 week training block. Treadmill grade was incorporated for two reasons: (1) to help the participants achieve maximal effort in the 30 s time frame, and (2) evidence suggests that exercising on a slight incline produces less orthopedic stress in overweight/obese individuals (
Ehlen et al. 2011). The researchers worked with the participants to select the maximal speed that participants could maintain for the full 30 s and that would elicit a maximal or near maximal HR response. To ensure that participants sprinted at maximal speed and grade for the full 30 s, researchers began to increase incline 15 s prior to the start of the sprint, and increased the speed 5 s prior to the start of the sprint. HR (using a Polar Heart Rate monitor) and RPE were recorded immediately after each sprint to ensure maximal effort. Speed and grade were adjusted as participants progressed throughout the 12 weeks to maintain HR and RPE. Sprints were paired with a 4 min walking recovery at 2.5 mph and 0% grade, with the final recovery consisting of a 5 min cool down. Every 3 weeks, the number of sprints was increased by two, with the final 3 weeks consisting of 10 maximal sprints.
MICT (
n = 7) participants walked or jogged continuously for 30 min on a treadmill at a 3–5% grade, at an intensity of their individual 45–55% HRR, which falls within the recommended range for moderate-intensity exercise (
ACSM 2013). A 5 min cool down at 2.5 mph and 0% grade completed each session. Every 3 weeks, the exercise session increased by 10 min, with the final 3 week block consisting of a 60 min sessions.
The total time commitment (i.e., time spent in the lab) was matched between groups; however, training volume differed between the groups (
Table 1). By the end of 12 weeks, the SIT group was obtaining 15 min of vigorous physical activity per week, and the MICT group was obtaining 180 min of moderate-intensity physical activity per week. Exercise adherence was quantified as the number of sessions attended out of 36 possible sessions.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics 21.0 (Armonk, New York, USA). The Levene’s test for normality was run and analyzed. A mixed-design ANOVA was used to compare all variables between groups and across time, with the exception of exercise adherence which was analyzed using an independent samples t test. In addition, we analyzed variables per protocol (SIT and MICT). Variables that were deemed significant were further investigated using independent samples t tests for measures between groups and paired-samples t tests for measures within groups. Significance was set a priori at p < 0.05.
Discussion
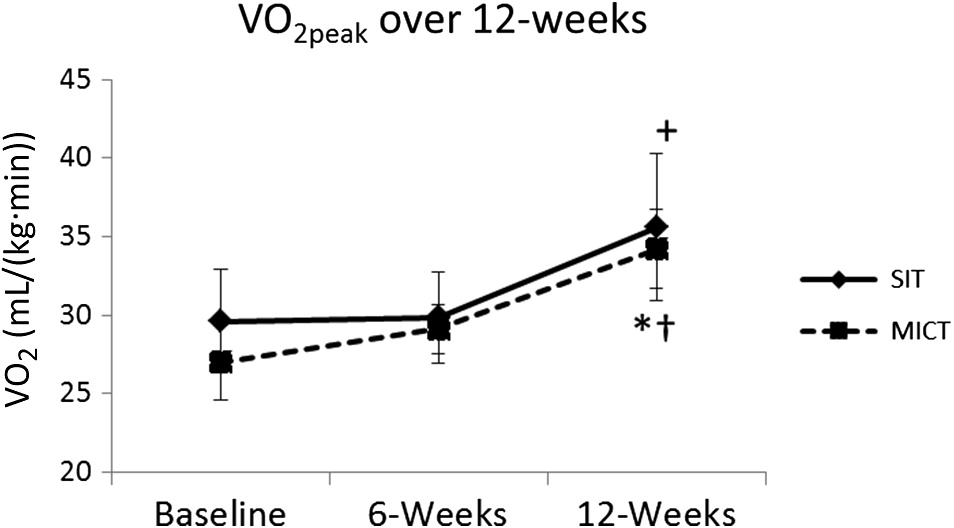
The purpose of this study was to examine the effects on cardiovascular disease risk factors of a 12 week run SIT protocol compared with that of a MICT protocol in a sample of healthy inactive, overweight/obese women, and to examine differences in perceived enjoyment and session RPE between the training protocols. This study demonstrated similar improvements in VO2peak and body composition in both groups over 12 weeks of training, despite a significantly lower training volume in the SIT group. Finally, despite a high-session RPE in the SIT group, participants reported similarly high perceived enjoyment levels as MICT, indicating that the increased intensity of the SIT protocol did not decrease enjoyment.
VO
2peak significantly increased by 20.7% in the SIT group despite the lower training volume. This finding is consistent with previous cycling SIT studies (
Burgomaster et al. 2005,
2008;
Whyte et al. 2010;
Bayati et al. 2011;
Astorino et al. 2012) and previous run SIT studies (
Macpherson et al. 2011;
Sandvei et al. 2012;
Hazell et al. 2014); however, this is the first study to examine the effects of run SIT on cardiorespiratory fitness in the overweight/obese population with poor aerobic capacity on an electric motor treadmill. These findings support the evidence that run SIT produces similar aerobic benefits to those of traditional cycling SIT and MICT. Moreover, the magnitude of improvement in VO
2peak for the SIT group in this study was slightly higher than those reported in previous run SIT studies (a ∼21% increase versus a ∼10% increase) (
Macpherson et al. 2011;
Sandvei et al. 2012;
Hazell et al. 2014). A recent study by
Gillen et al. (2016) reported similar increases in VO
2max (∼20%) after 12 weeks of cycle SIT training in sedentary men. It is speculated that changes in oxygen consumption occur in the periphery, with adaptations occurring at the point of gas exchange within the muscle (
Gist et al. 2014). It has also been postulated that adaptations within central command are causing the increase in VO
2max (
Sloth et al. 2013). Still, the dramatic changes observed in the current study could be due in part to the longer intervention design and (or) lower baseline fitness levels of our participants, as rapid improvements are more likely to occur in inactive, unfit individuals (
Bassett and Howley 2000).
It should be noted that the SIT group did not experience any significant improvement in VO
2peak during the first 6 weeks. This may indicate the presence of a critical threshold in terms of number of sprints per session that will elicit changes in aerobic capacity when using a motor-driven treadmill, similar to the dose–response relationship experienced with traditional moderate-intensity exercise (
Oja 2001). Alternatively, perhaps the sprint bout length was not sufficient to elicit changes in maximal oxygen consumption. This finding is contrary to the evidence presented by
Hazell et al. (2014) and
Macpherson et al. (2011) and previous cycle SIT studies (
Astorino et al. 2011;
Trilk et al. 2011;
Whyte et al. 2010), which report improvement in aerobic capacity within a 6 week time frame, despite the same number of sprints in this time block.
To our knowledge, this is the first study to demonstrate that an SIT protocol using a motor-driven treadmill can be effective for fat loss in women who were previously inactive and overweight/obese. The SIT group experienced a significant 1.7% decrease in total body fat percentage over the 12 week intervention, without a targeted dietary intervention, which supports previous SIT studies which were of shorter duration (
Macpherson et al. 2011;
Hazell et al. 2014). It should be noted that both
Hazell et al. (2014) and
Macpherson et al. (2011) used a treadmill set at dynamic mode which makes the participant generate all of the power, which could potentially elicit more rapid physiological changes compared to the electric motor treadmill used in the present study. Our results are similar to those of
Sijie et al. (2012) and
Heydari et al. (2012), who reported significant decreases in total body fat after 12 weeks of HIIT in sedentary, overweight adults. However, similar research in sedentary, normal weight adults reported no changes in body composition (
Astorino et al. 2013); however, it should be noted that this group used skinfold technique to assess body composition.
Further, the mechanisms of fat loss during an SIT intervention are not fully understood. Increased fat oxidation at rest and during exercise was reported after 2 weeks of HIIT training and could impact fat metabolism (
Talanian et al. 2007; Whyte et al. 2010). In addition, numerous SIT studies reported substantial increases in skeletal muscle enzymes, typically associated with endurance training, that play a key role in fat metabolism (
MacDougall et al. 1998;
Burgomaster et al. 2008;
Perry et al. 2008;
Whyte et al. 2013). Moreover, increased excess post exercise oxygen consumption (EPOC) following SIT sessions likely contributes to our observed improvement in body composition (
Hazell et al. 2012;
Chan and Burns 2013), despite the low training volume. Two studies have shown that HIIT/SIT training can induce significant levels of EPOC relative to a traditional endurance training group (
Skelly et al. 2014), regardless of sex or training modality (
Townsend et al. 2014).
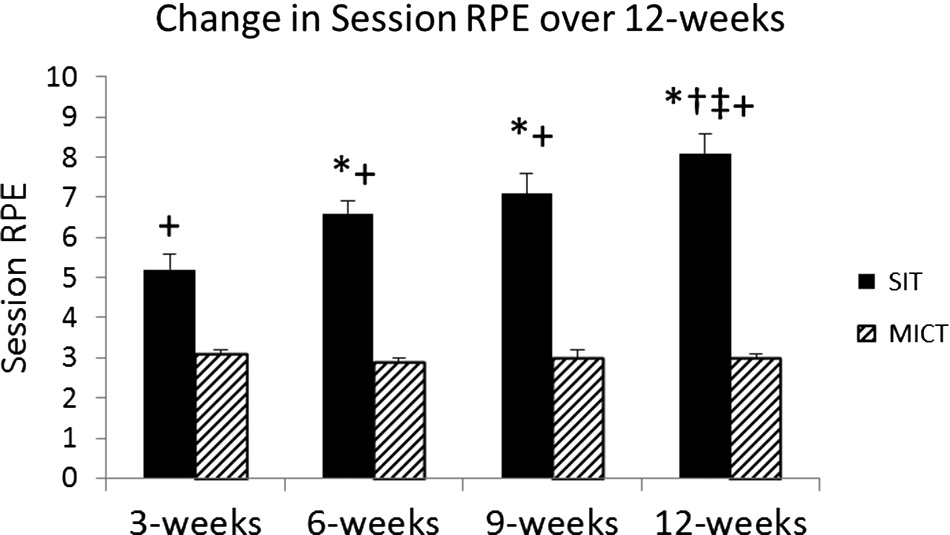
As previously mentioned, participation in regular physical activity in the overweight/obese population is lacking. In the present study, both the SIT and MICT groups reported high perceived enjoyment at baseline and throughout the intervention, despite the significantly higher session RPE in the SIT group. Thus, in an overweight/obese population, intense, vigorous activity with regular breaks was perceived as equally enjoyable as moderate-intensity walking/jogging. This finding differs from the findings of
Bartlett et al. (2011) who reported significantly higher enjoyment after an acute bout of HIIT compared to MICT. However, given our smaller sample size it is possible that the SIT group would have reported significantly greater enjoyment given a larger sample size. Anecdotally, a number of participants in the SIT group reported enjoying the sprint protocol because “
Anyone can do anything for 30-seconds”, and “
I feel like I am getting a really good workout even with the breaks in between [sprints]”. In addition, many of the participants commented on a desire to have a regular exercise companion, which is consistent with previous findings that reported women tend to desire more social support to physical activity (
Speck and Harrell 2003;
Hankonen et al. 2010). Together, these findings indicate that an SIT protocol would be an appropriate addition to an exercise program for overweight/obese women, particularly in a setting in which they had a companion they could talk to during the breaks.
A number of the investigated variables did not change significantly over the 12 week intervention. A non-significant reduction in HbA1c was observed during the last 6 weeks of training in the SIT group (
p = 0.141), and there was a near-significant decrease in TG over 12 weeks (
p = 0.056). The absence of significant improvement in blood lipids, blood glucose, and HbA1c is likely due to the fact that all participants had relatively healthy levels at baseline, despite being inactive and overweight. Previous literature on the effect of SIT on blood lipid profiles is inconclusive (
Sandvei et al. 2012;
Hazell et al. 2014). Traditional aerobic training is generally recommended to help lower LDL-C and TG, increase HDL-C, and improve autonomic tone and unregulated glucose (
Donnelly et al. 2009), thus further research on both a longer intervention and a greater participant pool is necessary to understand SIT’s long-term effect on blood lipids and glucose regulation
There were a few limitations to this study. The first is the low number of participants. As stated earlier, there were three dropouts in the SIT group, which may indicate a polarizing effect of the protocol itself that either attracts or deters individuals. It should be noted, however, that the majority of the participants were able to successfully complete the 12 week training protocol. The treadmill incline served to improve shock absorption (
Ehlen et al. 2011) and increase stability by allowing participants to decrease walking/running speed while still eliciting maximal effort. The second limitation is the intentional social support provided by the researchers. There appears to be a strong connection between increased physical activity levels in women and high levels of social support (
Speck and Harrell 2003); however, this type of opportunity does not always present itself naturally. It is possible that high levels of social support are what led to high levels of reported enjoyment, thus raising the question of whether the participants actually enjoyed the exercise protocol as opposed to simply enjoying the exercise setting. The third limitation stems from the lack of anticipated HR response in the SIT participants. Despite the researcher’s attempt to elicit a supramaximal HR response in the SIT group, approximately 50% of the recorded HR’s fell below 90% age-predicted HR
max, and the other 50% fell within 90–99% HR
max. Perhaps this is a limitation associated with the selected modality despite the participant’s perceived maximal exertion. Still, SIT studies that use manual powered treadmills are subject to individual effort and motivation which can vary from session to session. A benefit of the motor-driven treadmill modality is that speed is guaranteed for the full 30 s. Finally, this sample was limited to inactive, overweight/obese women who were otherwise healthy. There is room for future research in more clinical populations to see if a run SIT protocol elicits similar results, and if those subjects can endure the intensity of the protocol.