Characterization of microplastics and anthropogenic fibers in surface waters of the North Saskatchewan River, Alberta, Canada
Abstract
Microplastics are globally ubiquitous contaminants, but quantitative data on their presence in freshwater environments are sparse. This study investigates the occurrence, composition, and spatial trends of microplastic contamination in the North Saskatchewan River flowing through Edmonton, Alberta, the fifth largest city in Canada. Surface water samples were collected from seven sites throughout the city, upstream and downstream of the city, and near potential point sources (i.e., a wastewater treatment plant). Samples were spiked with fluorescent microbeads as internal standards and extracted by wet peroxide oxidation and density floatation. Microplastics were found in all samples, ranging in concentration from 4.6 to 88.3 particles·m−3 (mean = 26.2 ± 18.4 particles·m−3). Fibers were the dominant morphology recovered, and most were of anthropogenic origin and chemically identified as dyed cotton or polyester by Raman microspectroscopy. The majority of fragments were identified as polyethylene or polypropylene. No upstream to downstream differences were found in concentration, size distribution, or morphological composition suggesting nonpoint sources of microplastics to the river. This study represents one of the first investigations into the occurrence of microplastics in the freshwater environment in western Canada and will provide a baseline for future studies.
Introduction
Over the past several decades, the prevalence of plastic usage in society has increased dramatically, with a >600% increase in the quantity of plastics produced since 1975 (Jambeck et al. 2015). Much of the plastic produced is destined for use as packaging and is disposed of within a year of manufacturing (Geyer et al. 2017), leading to an increase in the quantity of plastic in solid municipal waste (Jambeck et al. 2015). This increased production, usage, and mismanagement of plastic waste has led to widespread global contamination of plastic pollution. Plastic is now a ubiquitous global contaminant found at both poles (Bergmann et al. 2017; Waller et al. 2017), in the deepest reaches of the ocean (Van Cauwenberghe et al. 2013), and in remote mountain regions (Free et al. 2014).
Microplastics are typically defined as plastics less than 5 mm and in the environment have potential deleterious effects on biota and ecosystem function. In particular, aquatic organisms ingest macro- and microplastics (Thompson 2004; Cole et al. 2013; Cole and Galloway 2015), which are transferred within food webs (Provencher et al. 2019). Ingestion of microplastics may cause physical effects on organisms, including abrasions, lacerations, or blockages of an organism’s digestive tract, resulting in a reduction in food intake, reduced growth, or death (Wright et al. 2013) as well as sub-lethal effects such as tissue inflammation, oxidative stress, and reduced locomotor behavior (Lu et al. 2016; Chen et al. 2017). Microplastics may also act as a vector for the transport and release of organic contaminants, such as pesticides, plasticizers, flame retardants, and additives, to organisms once ingested (Teuten et al. 2007, 2009). Because of the limited number of toxicology studies conducted using environmentally relevant exposures, the risk posed by microplastics to freshwater organisms and ecosystems is still unclear (Triebskorn et al. 2019).
Although most studies have focused on characterizing quantities of microplastics in the oceans, recent studies demonstrate that freshwater ecosystems are contaminated at similar or greater levels than marine environments. For example, an average of 43 000 particles·km−2 were found in the Great Lakes (Eriksen et al. 2013), which exceeds quantities found in open water marine environments (W.C. Li et al. 2016). Further, rivers are an important vector for transport of microplastics to the marine environment, and it is estimated that between 1.15 and 2.41 million tonnes of plastic is transported to the oceans via rivers annually (Lebreton et al. 2017). Globally, reported concentrations of microplastics in freshwater systems are variable and range over several orders of magnitude (Moore et al. 2011; Leslie et al. 2017; Lahens et al. 2018; Lin et al. 2018). Urbanization, in particular, is a key driver of microplastic pollution to freshwater systems (Peters and Bratton 2016; Kataoka et al. 2019). Important vectors for the introduction of microplastics to freshwater systems include the release of microplastics and synthetic fibers from wastewater treatment plants (WWTP; Mason et al. 2016; Gies et al. 2018), stormwater runoff (Grbić et al. 2020), runoff from agricultural fields with applied biosolids (Crossman et al. 2020), and atmospheric deposition (Dris et al. 2015a). Once introduced into the environment, little is unknown about the transport and fate of microplastics in freshwater systems.
Canada has approximately 20% of global surface freshwater, yet data on the occurrence of microplastics in freshwater ecosystems within Canada is lacking (Anderson et al. 2016). High levels of microplastic pollution have been reported in the Great Lakes (Eriksen et al. 2013; Corcoran et al. 2015; Dean et al. 2018; Mason et al. 2020) and Lake Winnipeg (Anderson et al. 2017), and in three river systems in Canada: the St. Lawrence river (Crew et al. 2020), the Ottawa River (Vermaire et al. 2017; Forrest et al. 2019), and the Red and Assiniboine Rivers (Warrack et al. 2017). Microplastic levels in these rivers are similar to those found in more populated river systems in Europe (Dris et al. 2015b). The occurrence of microplastic pollution in freshwater bodies in western Canada, however, is virtually unknown. We undertook the present study to address these knowledge gaps by quantifying and characterizing microplastics within an urbanized Canadian river. This study provides data on the prevalence of microplastic pollution within the Canadian freshwater environment needed to help understand the ecotoxicological risk posed by microplastics and provides baseline data of microplastic abundances on which future monitoring programs can be built.
Materials and methods
Study area
The North Saskatchewan River forms in the icefields of Banff and Jasper National Parks in Alberta and flows 1287 km eastward into Lake Winnipeg and eventually Hudson Bay. The total catchment area is approximately 122 800 km2 and includes most of southern and central Alberta and Saskatchewan. Edmonton, with a population of 930 000, is the second largest city in Alberta, the fifth largest city in Canada, and the largest city within the North Saskatchewan River watershed. It is the first major urban area encountered by the river, although several smaller municipalities and wastewater treatment plants are located upstream of the city.
Sample collection
Twenty-two surface water samples were collected in July and August 2017 from eight sites along an approximately 66 km stretch of the North Saskatchewan River (Fig. 1, Table S1). One site upstream of Edmonton (site 1) was included as a baseline site. Within the city limits, six sites were chosen, including sites in predominantly residential areas, throughout the downtown core, and one site each located immediately upstream (site 5) and downstream (site 6) of the municipal WWTP. A single sample was collected from each site in July, and duplicate samples were collected at each site in August, with the exception of site 3 (Emily Murphy) and site 7 (Hermitage), where only a single sample was collected in August.
Fig. 1.
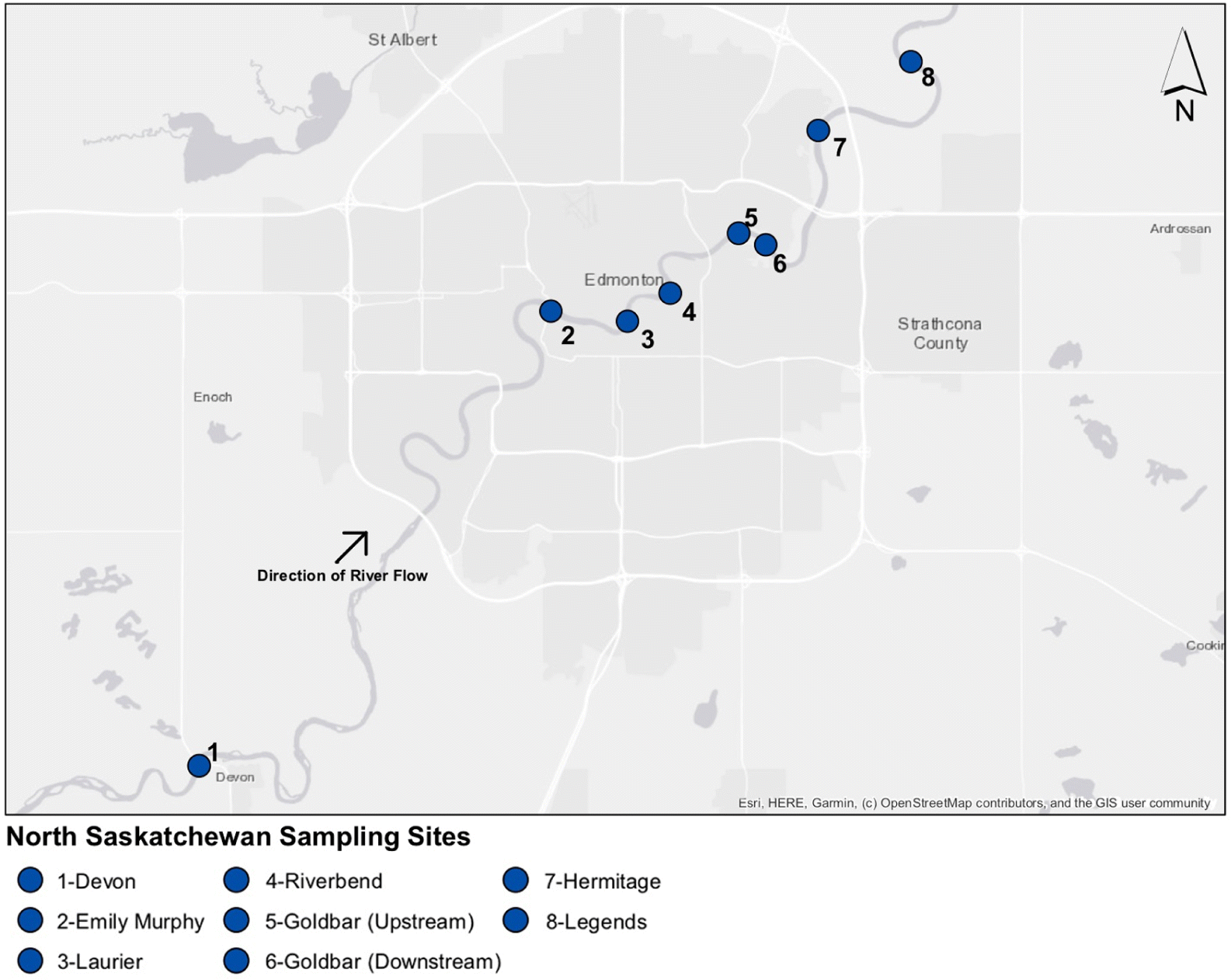
Sampling occurred near the shore by wading into the river. All samples were collected using an 8-inch nylon plankton net with 53 μm mesh (Wildco, Yulee, Florida, USA). Nets were fully submerged for 5 min into the flowing water just below the surface with the net opening facing the direction of water flow, enabling the collection of (on average) 5.5 m3 of water (Table S1). The nets were rinsed from the outside with deionized water that had previously been filtered with 0.22 μm filters to transfer the trapped particulates into the cod end of the net. The contents of the net were transferred into 1 L glass bottles and stored at 4 °C until analysis. At each site, river flow rates were measured using a PASPORT PS-2130 Flow Rate/Temperature Sensor (Pasco, Roseville, California, USA) immediately prior to sample collection, and depth was determined at the point of sample collection using a meter stick.
Microplastic quantities in one sample (site 8—July) were excluded from concentration data set due to unreliable measurements of water flow rates. However, the morphological and chemical identification data from this site are included in the analysis as sampling volume will not impact these percentages of these categories.
Sample processing
Prior to extraction, all samples and blanks were spiked with internal standards consisting of a known number of fluorescent polyethylene microbeads (Cospheric, Goleta, California, USA) of varying diameters and densities (Table S1) to assess the efficiency of the extraction procedure. We aimed to spike each sample at levels similar to the number of microplastics that may be expected; therefore, each sample was spiked with approximately 25–50 beads of each color. Following this, microplastics were isolated using density separation and wet peroxide oxidation (WPO) (Masura et al. 2015). All samples were filtered through a 37 μm stainless steel mesh sieve to isolate the solid particulate matter. The collected material was transferred to a 250 mL Erlenmeyer flask, loosely covered with rinsed aluminum foil and placed overnight in an oven at 70 °C to remove any residual water. Plastics were separated from denser material using a solution of ZnCl2 (ρ = 1.55–1.60 g·cm−3; VWR International, Mississauga, Ontario, Canada). Approximately 25 mL of the ZnCl2 solution was added to the flask and shaken on a shaker plate (Innova 44, New Brunswick Scientific, Edison, New Jersey, USA) at 125 rotations per minute (rpm) for 20 min to allow for mixing. The entire contents of the flask were then transferred to a density separation apparatus analogous to the one described by Masura et al. (2015). After settling overnight, the denser material was drained and discarded. The remaining material was filtered through a 37 μm stainless steel mesh sieve, rinsed well with filtered deionized water to remove any residual ZnCl2, and transferred to a clean Erlenmeyer flask. This flask was dried overnight at 70 °C to evaporate residual water. Samples then underwent digestion by WPO to remove organic materials and aid in the subsequent Raman analysis. Briefly, 20 mL of 30% H2O2 (Sigma Aldrich, Oakville, Ontario, Canada) and 20 mL of 0.05 M FeSO4 (Sigma Aldrich, Oakville, Ontario, Canada) were added to the flask and shaken on a shaker plate at 90 rpm and 50 °C for 60 min. If any organic material remained at the end of this time, an additional 20 mL of H2O2 was added, and reaction was continued for an additional 30 min.
Following removal of organic material, the contents of the flask were transferred to a sieve stack containing five stainless steel mesh sieves stacked on top of one another. The following mesh sizes were used: 37 μm, 125 μm, 250 μm, 500 μm, and 1 mm, resulting in separation of microplastics into the following size classes: >1 mm, 1 mm–500 μm, 500–250 μm, 250–125 μm, and 125–53 μm (due to the 53 μm net used during sampling). The sieve stack was rinsed thoroughly with filtered deionized water to remove any residual WPO reagents and aide in separation. The contents of each sieve were transferred individually with filtered deionized water to 0.45 μm gridded nitrocellulose filters (Ahlstrom-Munksjö, Helsinki, Finland), dried by vacuum filtration, and stored in an enclosed slide (Analyslide, Pall Laboratory, New York, USA) at 4 °C until enumeration and Raman analysis.
Quality control
During sampling, extraction, and analysis, care was taken to minimize contamination from external sources. Analysts wore cotton lab coats, and all glassware and laboratory utensils were washed thoroughly with filtered deionized water prior to use. All glassware was covered with aluminum foil throughout to minimize contamination from airborne plastics.
Laboratory blanks (n = 4), field blanks (n = 1), and equipment blanks (n = 1) were incorporated into the analysis scheme. Laboratory blanks consisted of 1 L of filtered (0.22 μm filter) deionized water. Field blanks were employed during each separate day of sample collection and consisted of 0.22 μm filtered deionized water in a 1 L glass bottle that was opened to the atmosphere during sample collection and subsequent transferring of the net contents. An equipment blank was used to assess potential contamination arising from carry-over between samples or from shedding of the nets themselves. The equipment blank was treated analogously to samples by thoroughly rinsing the outside of the net with filtered water, followed by transferring the contents of the net into 1 L glass bottles. All blanks were processed and analyzed identically to samples.
Suspected microplastics were found in all blanks, despite the precautions taken to minimize contamination. There were no differences in quantities of microplastic amongst blank types (i.e., field, net, and laboratory blanks), suggesting that the predominant source of contamination arose from the laboratory environment. Due to this lack of difference, all blanks were combined for further analyses. Following visual analysis, the number of identified microplastics in each sample was corrected for blank contamination by subtracting from each sample the average number of microplastics in each category (size and morphology) identified in the blanks. Uncorrected counts of microplastics identified in all samples and blanks can be found in Tables S2 and S3, respectively.
Visual analysis
Identification and quantification of microplastics was carried out by brightfield microscopy using either a stereo microscope (Olympus SZ61) or a compound microscope (Olympus CX41) under magnification ranging from 2× to 40×. Microplastics were identified based on previously established criteria (Hidalgo-Ruz et al. 2012). Briefly, suspected microplastics were required to be free of any cellular structures, be homogenous in color throughout, and fibers should be of consistent diameter throughout their entire length. Based on structural characteristics, all identified microplastics were placed into one of four categories: spheres (round and regularly shaped), films (irregularly shaped with similar length and width, but thin and flexible), fragments (length and width similar; irregularly shaped), or fibers (length greater than diameter). The color of each suspected microplastic was also recorded for July samples only and placed into one of the following categories: blue, red, clear, black, green, or other.
Chemical identification
Owing to time constraints, chemical identification of suspected microplastics was performed only on July samples. Raman microspectroscopy was carried out on 427 of the 773 visually identified microplastics (52.7%) across all sampling sites and blanks. Raman spectra of individual suspected microplastics and fibers were acquired with a SENTERRA I Raman microscope and Opus 6.5 software (Bruker Optics Ltd., Milton, Ontario, Canada). All spectra were acquired over the range of 45–3700 cm−1 using a 785 nm laser to minimize fluorescence. Typically, spectra were acquired with a laser intensity of 10 mW, a 5 s integration time, and five co-additions; however, these parameters were modified for some samples (e.g., fibers) to acquire stronger spectra or prevent decomposition of the sample. Prior to library matching, spectra were preprocessed by cutting from 45 to 200 cm−1 and 2500 to 3700 cm−1 to remove noisy regions of the spectra that contained no spectral information but hindered library matching. Spectra were further baseline corrected using a 65-point rubber band correction and min–max normalized. All preprocessing and library matching were done using Opus 6.5 software. The chemical composition of individual microplastics was obtained by matching acquired spectra to spectra contained in the SLoPP and SLoPP-E microplastic spectral libraries (Munno et al. 2020). To be considered a match a hit quality value of greater than 750 was required (where HQ = 1000 is a perfect match and 0 indicates no correlation at all), and all matches were visually confirmed to eliminate false positives. Samples that were not matched to any library spectra were classified as either “Unknown (Anthropogenic)” if they were colored (indicative of anthropogenic dye) or as “Unknown”.
Data analysis
All statistical tests were performed using GraphPad Prism version 8.3.0 software (GraphPad Software, La Jolla, California, USA). Parametric statistical tests were used when the appropriate assumptions were met and included t tests, one-way ANOVA followed by Tukey’s multiple comparisons test, and two-way ANOVA. Nonparametric Kruskal–Wallis tests followed by Dunn’s Multiple were performed on data sets that did not pass normality tests or did not meet the requirements for parametric tests. For all analyses, α = 0.05 was set as the level of significance. All data are presented as mean ± 1 standard deviation.
Results and discussion
Recoveries
To assess the efficiency of the extraction process, all samples and blanks were spiked with fluorescent microbeads in a range of sizes and densities (Table S4). The overall recovery of spiked fluorescent microbeads was 67.9% ± 34.9%, and mean recoveries ranging from 29.8% to 98.6%. Recoveries were dependent on size but not density (Table S4, Fig. S1) with larger microbeads (>150 μm) being extracted more efficiently than smaller beads (<150 μm, Kruskal–Wallis Test, H = 116.2, p < 0.001); the mean recovery of larger microbeads was 83.0% ± 26.5% compared with 44.7% ± 32.7% for smaller microbeads. We also found considerable differences in variability between size classes, with smaller particles having a relative standard deviation (RSD) of 73.25% compared with an RSD of 31.85% for larger particles (>150 μm). The low recoveries and greater variability for the smaller microbeads is likely due to adhesion of beads to the sides of the extraction vessels, density separation device, and (or) vacuum filtration apparatus. Although care was taken to rinse all glassware multiple times when transferring contents from one container to another, the fluorescent beads made it possible to visualize where some microbeads were located during the extraction process. The use of internal standards is widely used in analytical chemistry, but we were only able to find a single other study that utilized internal standards for assessment of microplastic recoveries (Zobkov and Esiukova 2017); in many studies, recoveries are assessed by external validation and spike and recovery. The inclusion of internal standards enables more reliable microplastic quantification within chosen size ranges on a sample by sample basis, and we encourage their inclusion in future studies. In an effort to be consistent with previous studies, we chose not to correct reported concentrations for internal standard recoveries. As a first assessment of their use, more work is also necessary to determine whether microbead recoveries are representative of other morphologies, such as fibers.
Total microplastic concentrations
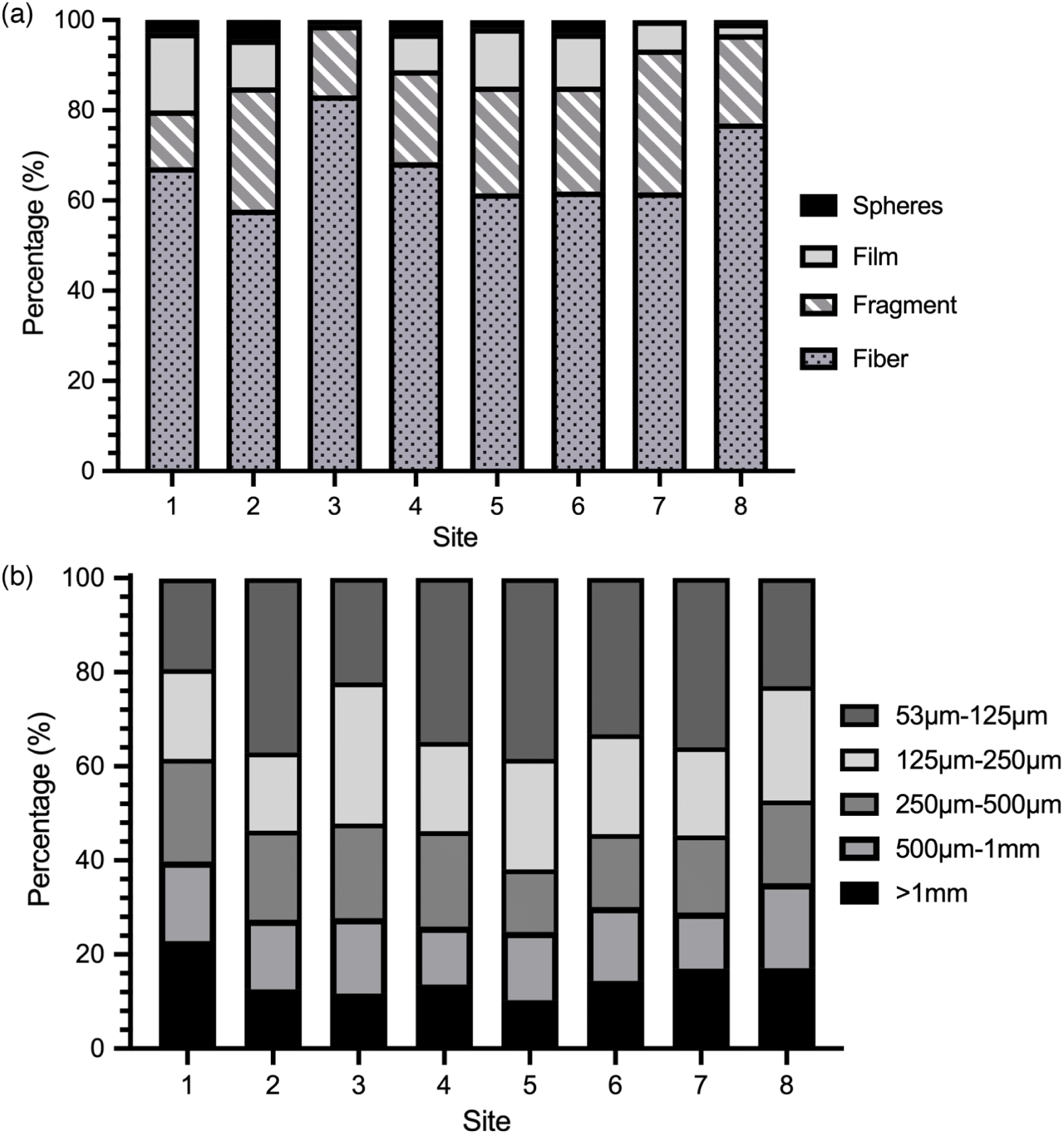
Microplastics and anthropogenic fibers were identified in all surface water samples, ranging in concentration from 4.6 to 88.3 particles·m−3, with a mean concentration (±1 standard deviation) across all samples of 26.2 ± 18.4 particles·m−3 (Fig. 2). Across all sites, fibers accounted for a majority (67.5% ± 8.7%) of the total microplastics in the North Saskatchewan River, followed by fragments (21.8% ± 6.1%), films (8.7% ± 5.5%), and spheres (2.0% ± 1.5%) (Fig. 3a). Total concentrations in August were higher than in July (Mann–Whitney test, U = 8, p = 0.0011; Fig. S2), although there were no differences in distribution of morphologies or sizes between sampling times.
Fig. 2.
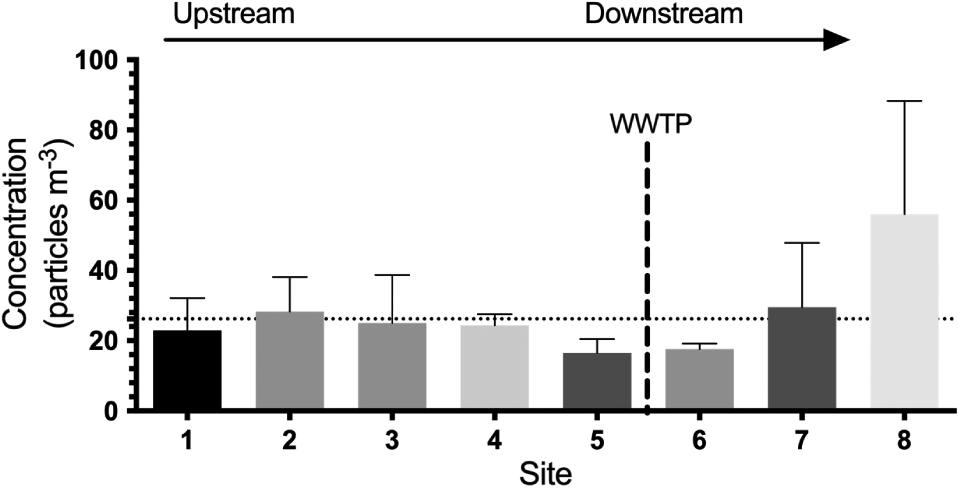
Fig. 3.
This is amongst the first studies to report on concentrations of microplastics in a western Canadian freshwater system and corroborates a multitude of previous findings demonstrating that microplastics are ubiquitous in freshwater environments. The microplastic concentrations reported herein are consistent with concentrations reported in other river systems in North America (McCormick et al. 2014; Baldwin et al. 2016; Estahbanati and Fahrenfeld 2016) and in particular, to concentrations of microplastics quantified in other river systems throughout Canada. Mean concentrations of microplastics in the North Saskatchewan River are higher than those reported in an urban creek downstream of Regina, Saskatchewan (population 229 000), (Campbell et al. 2017) and approximately five-fold higher than those reported for the Red, Assiniboine, and Nelson rivers, near Winnipeg, Manitoba (population 700 000), where microplastic concentrations averaged of 5.3 microplastics·m−3 (Warrack et al. 2017). Similar concentrations of 12 particles·m−3 and 16 particles·m−3 were found upstream and downstream of wastewater treatment plants, respectively, in the St. Lawrence River, despite also reporting amongst the highest sediment concentrations thus far reported for freshwater systems (Crew et al. 2020). Concentrations in the North Saskatchewan River are approximately four-fold lower than those reported by Vermaire et al. (2017) and Forrest et al. (2019) for the Ottawa river and its tributaries, which flow through Ottawa, Ontario, a city of comparable size to Edmonton (1.23 million vs. 900 000). In this system, concentrations ranged from 20 to 410 particles·m−3 based on 1 L grab samples. However, the concentrations reported herein are greater than the mean of 1.35 particles·m−3 reported for the Ottawa River and tributaries when water was sampled using high-volume manta trawls (Vermaire et al. 2017).
Comparisons of microplastics concentrations amongst studies is difficult, however, given differences sampling and analytical methodology utilized amongst studies. In particular, differences in total concentrations may be attributable to the difference in mesh sizes used for sampling. In this study, a 53 μm mesh size was chosen in an effort to capture the widest possible range of potential microplastics—smaller than the 333 μm mesh size used by Warrack et al. (2017) or the 100 μm filters used in studies of the Ottawa (Vermaire et al. 2017; Forrest et al. 2019) and St. Lawrence Rivers (Crew et al. 2020). Differences in mesh size have a substantial impact on the number of plastics recovered, as other studies have shown a 250-fold increase in the number of plastics captured following a decrease in mesh size from 333 to 80 μm (Dris et al. 2018). Moreover, the most prevalent microplastics found in the North Saskatchewan River were those in the smallest size range investigated (53–125 μm), which comprised 30.5% ± 7.7% of the total microplastics identified (Fig. 3b). This is consistent with other studies in both freshwater and marine environments that found concentrations of microplastics increase with decreasing particle size (Estahbanati and Fahrenfeld 2016; Lindeque et al. 2020). Although our findings are in line with these previous studies, it is germane to note that difficulty in positively identifying microplastics by light microscopy increases with decreasing size. At particles sizes <100 μm, rates of positive identification for visual identification fall below 80%, leading to an overestimate of the true particle counts within smaller size fractions (Lenz et al. 2015). We were unable to attempt Raman microspectroscopy on all particles in this size fraction; however, 71.8% of the particles in this size fraction in which we were able to successfully acquire a Raman spectrum were identified as either plastics or unknown anthropogenic, which was consistent with other size fractions examined in this study.
Considerable spatial and temporal heterogeneity in microplastic concentrations exist within dynamic river systems, and microplastic concentrations may vary with sampling depth or sampled location within the cross-section of the river (Dris et al. 2018; Forrest et al. 2019; Lenaker et al. 2019), further complicating comparisons amongst studies. All samples were collected from the near-shore environment, although other studies have sampled river channels from boats or bridges. Comparisons are also hindered by differences in sampling methodology, as grab sampling results in higher reported concentrations than bulk water sampling (Vermaire et al. 2017). Such reported differences makes clear the need for greater standardization in sampling methodology and (or) sampling under similar hydrogeomorphic river conditions to help to systematize microplastic sampling in rivers within riverine systems.
Overall concentrations varied temporally, with total concentrations across all sites being higher in August compared with July. Although mobilization and transport of land-based microplastics to the river following precipitation events have been identified as contributing to temporal changes in microplastic concentrations within in rivers (Dris et al. 2015b), this is unlikely to be the cause for the trends observed here, as the total precipitation amounts in Edmonton were similar in the 7 d prior to each sampling day (July: 2.8 mm, August: 2 mm) and no precipitation within 3 d of either sampling day. Rather, we observed both decreased depth and increased flow velocity at sampling sites in August compared with July (Fig. S3). The increased velocity and decreased depth may have led to the resuspension of settled microplastics from sediments. However, in addition to urban runoff dynamics, hydrogeomorphic flow of the river would change with both local and upstream precipitation events. With a large and dynamic river such as the North Saskatchewan river, daily and seasonal fluctuations in discharge may lead to local variability at different time points. Further sampling is required to confirm and further elucidate any temporal trends within the North Saskatchewan River.
Characterization of microplastics
Synthetic fibers have been widely reported as the primary microplastic morphology found in freshwater ecosystems globally (Dris et al. 2015b; Baldwin et al. 2016; Leslie et al. 2017; Lin et al. 2018), and the preponderance of fibers found in the North Saskatchewan River are consistent with these earlier reports. Moreover, this composition is similar to the percentage of fibers found in the Ottawa River and the Red and Assiniboine Rivers in Manitoba (Vermaire et al. 2017; Warrack et al. 2017; Forrest et al. 2019), Wascana Creek in Saskatchewan (Campbell et al. 2017), and Lake Winnipeg (Anderson et al. 2017).
Most of the fibers were either blue, clear, or black in color (Fig. S4) and were chemically identified as polyester or cotton (Fig. 4). We were unable to conclusively identify the chemical identity of 32% of the fibers due to the Raman spectrum of the polymer being obscured by that of the dyes, a common problem in Raman spectroscopy (Lenz et al. 2015). However, these unknown fibers were dyed and most likely of anthropogenic origin. Of these unknown fibers, most (62%) were blue in color and tentatively identified as containing indigo dyes, based on comparison with published spectra (Baran et al. 2010). Indigo dye is widely used in the dying of cotton fabric for the manufacturing of denim and use in blue jeans and has been identified as being major components of fiber pollution in wastewater treatment plant effluent (Mason et al. 2016). However, we found a dominance of fibers upstream of the WWTP, indicating the presence of other sources. These fibers may have originated from effluent discharged into the North Saskatchewan River from upstream WWTPs, although fibers also compose a large proportion of microplastics in stormwater runoff (Grbić et al. 2020) and urban air (Dris et al. 2015a; Wright et al. 2020), suggesting that these sources may also be contributing to the microplastic load to the North Saskatchewan River.
Fig. 4.
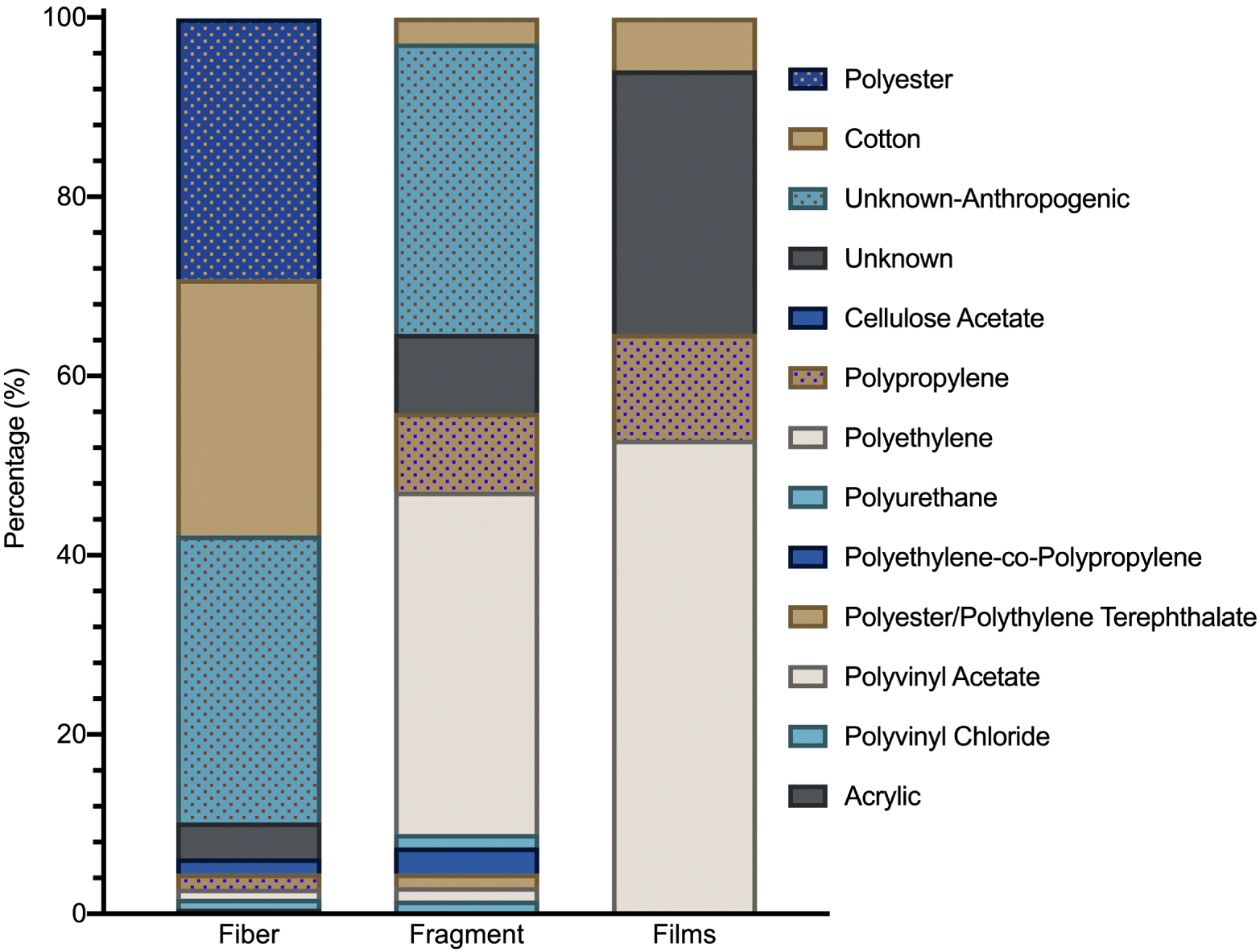
Most fragments found in this study were clear, with large proportions of blue, black, or red also present. Most (32.4%) were chemically identified as polyethylene or polypropylene (8.8%), consistent with other studies and with the high production volume of these two polymers (Geyer et al. 2017). Like fibers, however, a large proportion (32.4%) of fragments were identified as anthropogenic in origin, but the polymer type was not chemically identified due to a lack of matching spectrum in our library and the influence of dyes or additives on the Raman spectrum.
Changes in the predominant colors of microplastics found in samples collected from urban sites and those collected upstream and downstream of urban areas, along with a greater diversity of the colors identified in urban sites, suggests that urban areas contribute greater variability in plastics to rivers than nonurban areas (Peters and Bratton 2016). However, we were unable to find any differences between urban and nonurban sites with respect to predominant color or color variability, although this may be a consequence of collecting samples from only a single upstream nonurban site, as well as the constrained categories of colors that were identified. Further, changes in the discoloration and bleaching of plastics may occur due to weathering or due to our digestion procedure (J. Li et al. 2016), which may have contributed to the preponderance of clear fragments and fibers found. The reporting of color distributions is important for ecotoxicological risk assessments though, as a number of species of aquatic organisms likely selectively ingest microplastics based on color preference (Wright et al. 2013).
Potential sources
Quantities of microplastics have been shown to be related to population densities and input from urban sources (Yonkos et al. 2014; Baldwin et al. 2016). We hypothesized, therefore, that microplastic concentrations would increase as the North Saskatchewan River flowed through the city of Edmonton. With the exception of site 8, the most downstream site, there were no significant differences in total concentrations of microplastics between sampling locations (Kruskal–Wallis test, H = 3.905, p = 0.79, df = 7) and no identifiable upstream to downstream trends (Fig. 2). Although a single WWTP outfall is located within the stretch of river sampled, we found no difference in concentrations, or any other investigated characteristics, between sites immediately upstream of the WWTP (site 5) and samples collected immediately downstream of the WWTP (site 6) (Fig. 2), suggesting that the WWTP is not acting as a significant point source of microplastic pollution to the river. The higher observed concentration of microplastics at the furthest downstream site (site 8), with a total microplastic concentration approximately 3.5-fold higher than other sites, may be due to the lower river flow rates at this site. Although there was no relationship between flow rate and concentration (r2 = 0.0199, p = 0.6160), flow rates at site 8 were the lowest of all of the sampling sites in this study (Table S1) and 30% lower than the next lowest site. Although point bars and regions of decreased river velocity lead to a depositional environment for microplastics and lower water column concentrations (Yonkos et al. 2014), the lower flow rate at site 8 may create a temporary accumulation zone for neutrally or positively buoyant microplastics prior to deposition. We intend to carry out further studies to elucidate the reason for the elevated concentrations at this site.
The finding that the WWTP is likely not acting as a point source for microplastic input to the river contrasts other studies, including studies in Canada, which found that local WWTPs were discharging microplastics to local waterways and that differences in concentration and profile were identifiable between upstream and downstream sampling locations (McCormick et al. 2014; Baldwin et al. 2016; Estahbanati and Fahrenfeld 2016; Vermaire et al. 2017; Warrack et al. 2017), although other studies have similarly reported a lack of influence from WWTPs (Hoellein et al. 2017; Crew et al. 2020). Such differences amongst studies may be a result of different treatment processes utilized at the various WWTPs (Talvitie et al. 2017) or sampled location relative to the outfall. The WWTP in Edmonton utilizes tertiary treatment (EPCOR 2020a), a process which removes >98% of the microplastic and fiber load that enters the treatment plant (Murphy et al. 2016). Thus, while microplastics and fibers are likely being discharged into the river, the total loadings to the river may not be sufficiently high to elicit a noticeable change in concentrations or morphological distribution at downstream sites. Alternatively, the choice of sampling location relative to the WWTP outfall may also impact reported concentrations. As microplastics will be carried toward the center of the river due to momentum from the WWTP effluent discharge, they will not be immediately mixed with the slower moving water near the shoreline. We sampled near the shore and within 100 m of the WWTP outfall, and at this close proximity to the outfall particles and fibers may not have mixed fully with the nearshore water (Pilechi et al. 2012). At the intermittent downstream site (site 7), samples were collected approximately 8.5 km downstream from the outfall and from the bank opposite from the WWTP outfall. At this distance downstream, the contaminant plume may not yet have fully transversed the river, or only the edges of the contaminant plume had reached the far bank (Pilechi et al. 2015). Thus, we may not have been able to detect any input from the WWTP. As discussed above, more work is necessary to standardize sampling within rivers, and the mixing of microplastics within a riverine system following discharge from WWTPs or stormwater outfalls requires additional study.
In addition to concentration, there was no significant variation in morphological composition (Fig. 3a), size distribution (Fig. 3b), color (Fig. S4), or chemical identity amongst sites (Fig. 4). Such similarity in characteristics amongst sites suggests that microplastics are being introduced to the North Saskatchewan River through diffuse, nonpoint sources. Microplastics and anthropogenic fibers found in the North Saskatchewan River likely originate from terrestrial sources and are introduced to the river via runoff or atmospheric transport and deposition. Like many jurisdictions located along rivers, stormwater runoff from the city of Edmonton enters the river untreated from multiple stormwater outflow pipes throughout the city. Stormwater runoff may contain concentrations of microplastics and fibers at levels comparable with or exceeding those found in treated WWTP effluent (Grbić et al. 2020), and thus outfalls may act as discrete point sources of microplastic pollution (Horton et al. 2017). There are approximately 120 stormwater outfalls that discharge into the North Saskatchewan River between sites 1 and 8 (EPCOR 2020b) and, despite being discrete point sources, may act collectively as a diffuse nonpoint source of pollutants to the river. Additionally, much of the land area along the North Saskatchewan river upstream of Edmonton and in the greater Edmonton region is used for agriculture. In Alberta, biosolids, which are formed from the solids retained in WWTPs (e.g., Goldbar WWTP is Edmonton’s largest and produces an average of 20 800 tonnes dry weight per year), are nutrient rich and are used as fertilizer for agricultural fields. Such biosolids retain the microplastics and fibers, which may be released to the environment through erosion and runoff (Crossman et al. 2020). Finally, microplastics are present in urban air and may be transported and deposited regionally in the environment surrounding urban areas (Wright et al. 2020). Morphological profiles were dominated by fibers (Wright et al. 2020), similar to our findings for North Saskatchewan River surface water, suggesting that atmospheric transport and deposition from the City of Edmonton may also be contributing to the microplastic and fiber load within the river.
Conclusions
Although reports of microplastics in freshwater environments are increasing, the field is generally still in its infancy. This study adds to the growing knowledge of microplastic occurrence in Canadian freshwater ecosystems and is the first, to our knowledge, to characterize microplastic pollution in a western Canadian river. Microplastics and other anthropogenic fibers were found in every sample, with the overall composition dominated by fibers and smaller microplastics. Similarities in concentration, morphologies, and chemical compositions amongst sites suggest diffuse sources of microplastics to the river. In addition to vertical and horizontal hydrogeomorphic characteristics of lotic systems, wind, biofouling, and interaction with other materials may affect the behavior of microplastics in rivers, possibly biasing sampling and obfuscating sources. More research is needed to understand how different sampling, extraction, and analytical methodologies may bias quantitative results, along with a greater understanding of how urban sources and riverine flow dynamics impact observed profiles and concentrations of microplastics.
Acknowledgements
Thank you to the anonymous reviewers for taking their time to review our manuscript and provide helpful comments that contributed to the improvement of this manuscript. The authors would also like to thank Dr. Haley Carlson for assistance in field sample collection and Tianna Groeneveld for help with the creation of the map. Funding was provided by MacEwan University and from the Natural Science and Engineering Research Council of Canada through a Discovery grant to MSR (funding reference number RGPIN-2015-04852).
References
Anderson JC, Park BJ, and Palace VP. 2016. Microplastics in aquatic environments: implications for Canadian ecosystems. Environmental Pollution, 218: 269–280.
Anderson PJ, Warrack S, Langen V, Challis JK, Hanson ML, and Rennie MD. 2017. Microplastic contamination in Lake Winnipeg, Canada. Environmental Pollution, 225: 223–231.
Baldwin AK, Corsi SR, and Mason SA. 2016. Plastic debris in 29 Great Lakes tributaries: relations to watershed attributes and hydrology. Environmental Science & Technology, 50(19): 10377–10385.
Baran A, Fiedler A, Schulz H, and Baranska M. 2010. In situ Raman and IR spectroscopic analysis of indigo dye. Analytical Methods, 2(9): 1372.
Bergmann M, Wirzberger V, Krumpen T, Lorenz C, Primpke S, Tekman MB, et al. 2017. High quantities of microplastic in arctic deep-sea sediments from the HAUSGARTEN observatory. Environmental Science & Technology, 51(19): 11000–11010.
Campbell SH, Williamson PR, and Hall BD. 2017. Microplastics in the gastrointestinal tracts of fish and the water from an urban prairie creek. FACETS, 2(1): 395–409.
Chen Q, Gundlach M, Yang S, Jiang J, Velki M, Yin D, et al. 2017. Quantitative investigation of the mechanisms of microplastics and nanoplastics toward zebrafish larvae locomotor activity. Science of The Total Environment, 584–585: 1022–1031.
Cole M, and Galloway TS. 2015. Ingestion of nanoplastics and microplastics by pacific oyster larvae. Environmental Science & Technology, 49(24): 14625–14632.
Cole M, Lindeque P, Fileman E, Halsband C, Goodhead R, Moger J, et al. 2013. Microplastic ingestion by zooplankton. Environmental Science & Technology, 47(12): 6646–6655.
Corcoran PL, Norris T, Ceccanese T, Walzak MJ, Helm PA, and Marvin CH. 2015. Hidden plastics of Lake Ontario, Canada and their potential preservation in the sediment record. Environmental Pollution, 204: 17–25.
Crew A, Gregory-Eaves I, and Ricciardi A. 2020. Distribution, abundance, and diversity of microplastics in the upper St. Lawrence River. Environmental Pollution, 260: 113994.
Crossman J, Hurley RR, Futter M, and Nizzetto L. 2020. Transfer and transport of microplastics from biosolids to agricultural soils and the wider environment. Science of The Total Environment, 724: 138334.
Dean BY, Corcoran PL, and Helm PA. 2018. Factors influencing microplastic abundances in nearshore, tributary and beach sediments along the Ontario shoreline of Lake Erie. Journal of Great Lakes Research, 44(5): 1002–1009.
Dris R, Gasperi J, Saad M, Cile Mirande C, and Tassin B. 2015a. Synthetic fibers in atmospheric fallout: a source of microplastics in the environment? Marine Pollution Bulletin, 104(1–2): 290–293.
Dris R, Gasperi J, Rocher V, Saad M, Renault N, and Tassin B. 2015b. Microplastic contamination in an urban area: a case study in Greater Paris. Environmental Chemistry, 12(5): 592–599.
Dris R, Gasperi J, Rocher V, and Tassin B. 2018. Synthetic and non-synthetic anthropogenic fibers in a river under the impact of Paris Megacity: sampling methodological aspects and flux estimations. Science of The Total Environment, 618: 157–164.
EPCOR. 2020a. Gold bar wastewater treatment process [online]: Available from epcor.com/about/who-we-are/where-we-operate/edmonton/Documents/GoldbarTreatmentProcess.pdf.
EPCOR. 2020b. City of edmonton open data: drainage-outfalls [online]: Available from data.edmonton.ca/Externally-Sourced-Datasets/Drainage-Outfall/hqkz-aab4.
Eriksen M, Mason S, Wilson S, Box C, Zellers A, Edwards W, et al. 2013. Microplastic pollution in the surface waters of the Laurentian Great Lakes. Marine Pollution Bulletin, 77(1–2): 177–182.
Estahbanati S, and Fahrenfeld NL. 2016. Influence of wastewater treatment plant discharges on microplastic concentrations in surface water. Chemosphere, 162: 277–284.
Forrest SA, Holman L, Murphy M, and Vermaire JC. 2019. Citizen science sampling programs as a technique for monitoring microplastic pollution: results, lessons learned and recommendations for working with volunteers for monitoring plastic pollution in freshwater ecosystems. Environmental Monitoring and Assessment, 191(3): 172.
Free CM, Jensen OP, Mason SA, Eriksen M, Williamson NJ, and Boldgiv B. 2014. High-levels of microplastic pollution in a large, remote, mountain lake. Marine Pollution Bulletin, 85(1): 156–163.
Geyer R, Jambeck JR, and Law KL. 2017. Production, use, and fate of all plastics ever made. Science Advances, 3(7): e1700782.
Gies EA, LeNoble JL, Noël M, Etemadifar A, Bishay F, Hall ER, et al. 2018. Retention of microplastics in a major secondary wastewater treatment plant in Vancouver, Canada. Marine Pollution Bulletin, 133: 553–561.
Grbić J, Helm P, Athey S, and Rochman CM. 2020. Microplastics entering northwestern Lake Ontario are diverse and linked to urban sources. Water Research, 174: 115623.
Hidalgo-Ruz V, Gutow L, Thompson RC, and Thiel M. 2012. Microplastics in the marine environment: a review of the methods used for identification and quantification. Environmental Science & Technology, 46(6): 3060–3075.
Hoellein TJ, McCormick AR, Hittie J, London MG, Scott JW, and Kelly JJ. 2017. Longitudinal patterns of microplastic concentration and bacterial assemblages in surface and benthic habitats of an urban river. Freshwater Science, 36(3): 491–507.
Horton AA, Svendsen C, Williams RJ, Spurgeon DJ, and Lahive E. 2017. Large microplastic particles in sediments of tributaries of the River Thames, UK—abundance, sources and methods for effective quantification. Marine Pollution Bulletin, 114(1): 218–226.
Jambeck JR, Geyer R, Wilcox C, Siegler TR, Perryman M, Andrady A, et al. 2015. Marine pollution. Plastic waste inputs from land into the ocean. Science (New York, N.Y.), 347(6223): 768–771.
Kataoka T, Nihei Y, Kudou K, and Hinata H. 2019. Assessment of the sources and inflow processes of microplastics in the river environments of Japan. Environmental Pollution, 244: 958–965.
Lahens L, Strady E, Kieu-Le T-C, Dris R, Boukerma K, Rinnert E, et al. 2018. Macroplastic and microplastic contamination assessment of a tropical river (Saigon River, Vietnam) transversed by a developing megacity. Environmental Pollution, 236: 661–671.
Lebreton LCM, van der Zwet J, Damsteeg J-W, Slat B, Andrady A, and Reisser J. 2017. River plastic emissions to the world’s oceans. Nature Communications, 8: 1–10.
Lenaker PL, Baldwin AK, Corsi SR, Mason SA, Reneau PC, and Scott JW. 2019. Vertical distribution of microplastics in the water column and surficial sediment from the Milwaukee River Basin to Lake Michigan. Environmental Science & Technology, 53(21): 12227–12237.
Lenz R, Enders K, Stedmon CA, MacKenzie DMA, and Nielsen TG. 2015. A critical assessment of visual identification of marine microplastic using Raman spectroscopy for analysis improvement. Marine Pollution Bulletin, 100(1): 82–91.
Leslie HA, Brandsma SH, van Velzen MJM, and Vethaak AD. 2017. Microplastics en route: field measurements in the Dutch river delta and Amsterdam canals, wastewater treatment plants, North Sea sediments and biota. Environment International, 101(C): 133–142.
Li J, Qu X, Su L, Zhang W, Yang D, Kolandhasamy P, Li D, and Shi H. 2016. Microplastics in mussels along the coastal waters of China. Environmental Pollution, 214: 177–184.
Li WC, Tse HF, and Fok L. 2016. Plastic waste in the marine environment: a review of sources, occurrence and effects. Science of The Total Environment, 566–567: 333–349.
Lin L, Zuo L-Z, Peng J-P, Cai L-Q, Fok L, Yan Y, et al. 2018. Occurrence and distribution of microplastics in an urban river: a case study in the Pearl River along Guangzhou City, China. Science of The Total Environment, 644: 375–381.
Lindeque PK, Cole M, Coppock RL, Lewis CN, Miller RZ, Watts AJR, et al. 2020. Are we underestimating microplastic abundance in the marine environment? A comparison of microplastic capture with nets of different mesh-size. Environmental Pollution, 265: 114721.
Lu Y, Zhang Y, Deng Y, Jiang W, Zhao Y, Geng J, et al. 2016. Uptake and accumulation of polystyrene microplastics in zebrafish (Danio rerio) and toxic effects in liver. Environmental Science & Technology, 50(7): 4054–4060.
Mason SA, Garneau D, Sutton R, Chu Y, Ehmann K, Barnes J, et al. 2016. Microplastic pollution is widely detected in US municipal wastewater treatment plant effluent. Environmental Pollution, 218(C): 1045–1054.
Mason SA, Daily J, Aleid G, Ricotta R, Smith M, Donnelly K, et al. 2020. High levels of pelagic plastic pollution within the surface waters of Lakes Erie and Ontario. Journal of Great Lakes Research, 46(2): 277–288.
Masura J, Baker J, Foster G, and Arthur C. 2015. Laboratory methods for the analysis of microplastics in the marine environment. (NOAA technical memorandum NOS-OR&R-48) [online]: Available from marinedebris.noaa.gov/technical-memorandum/laboratory-methods-analysis-microplastics-marine-environment.
McCormick A, Hoellein TJ, Mason SA, Schluep J, and Kelly JJ. 2014. Microplastic is an abundant and distinct microbial habitat in an Urban River. Environmental Science & Technology, 48: 11863–11871.
Moore CJ, Lattin GL, and Zellers AF. 2011. Quantity and type of plastic debris flowing from two urban rivers to coastal waters and beaches of Southern California. Revista de Gestão Costeira Integrada, 11(1): 65–73.
Munno K, De Frond H, O’Donnell B, and Rochman CM. 2020. Increasing the accessibility for characterizing microplastics: introducing new application-based and spectral libraries of plastic particles (SLoPP and SLoPP-E). Analytical Chemistry, 92(3): 2443–2451.
Murphy F, Ewins C, Carbonnier F, and Quinn B. 2016. Wastewater treatment works (WwTW) as a source of microplastics in the aquatic environment. Environmental Science & Technology, 50(11): 5800–5808.
Peters CA, and Bratton SP. 2016. Urbanization is a major influence on microplastic ingestion by sunfish in the Brazos River Basin, Central Texas, USA. Environmental Pollution, 210(c): 380–387.
Pilechi A, Rennie CD, Mohammadian M, Zhu D, and Delatolla R. 2012. Physical mixing patterns of water and contaminants in the North Saskatchewan River. [online]: Available from open.alberta.ca/dataset/d633b40c-04b7-468d-bd10-4252e52ef987/resource/5f3afe17-207d-4a8b-b1f6-a451de3b9423/download/8900.pdf.
Pilechi A, Rennie CD, Mohammadian A, and Zhu DZ. 2015. In situ spatially distributed field measurements of transverse dispersion of a wastewater effluent in an extended natural meandering river. Journal of Hydraulic Research, 53(1): 20–35.
Provencher JF, Ammendolia J, Rochman CM, and Mallory ML. 2019. Assessing plastic debris in aquatic food webs: what we know and don’t know about uptake and trophic transfer. Environmental Reviews, 27(3): 304–317.
Talvitie J, Mikola A, Koistinen A, and Setälä O. 2017. Solutions to microplastic pollution—removal of microplastics from wastewater effluent with advanced wastewater treatment technologies. Water Research, 123: 401–407.
Teuten EL, Rowland SJ, Galloway TS, and Thompson RC. 2007. Potential for plastics to transport hydrophobic contaminants. Environmental Science & Technology, 41(22): 7759–7764.
Teuten EL, Saquing JM, Knappe DRU, Barlaz MA, Jonsson S, Björn A, et al. 2009. Transport and release of chemicals from plastics to the environment and to wildlife. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 364(1526): 2027–2045.
Thompson RC. 2004. Lost at sea: where is all the plastic? Science (New York, N.Y.), 304(5672): 838.
Triebskorn R, Braunbeck T, Grummt T, Hanslik L, Huppertsberg S, Jekel M, et al. 2019. Relevance of nano- and microplastics for freshwater ecosystems: a critical review. TrAC Trends in Analytical Chemistry, 110: 375–392.
Van Cauwenberghe L, Vanreusel A, Mees J, and Janssen CR. 2013. Microplastic pollution in deep-sea sediments. Environmental Pollution, 182(C): 495–499.
Vermaire JC, Pomeroy C, Herczegh SM, Haggart O, and Murphy M. 2017. Microplastic abundance and distribution in the open water and sediment of the Ottawa River, Canada, and its tributaries. FACETS, 2(1): 301–314.
Waller CL, Griffiths HJ, Waluda CM, Thorpe SE, Loaiza I, Moreno B, et al. 2017. Microplastics in the Antarctic marine system: an emerging area of research. Science of The Total Environment, 598: 220–227.
Warrack S, Challis JK, Hanson ML, and Rennie MD. 2017. Microplastics flowing into Lake Winnipeg: densities, sources, flux, and fish exposures. Proceedings of Manitoba’s Undergraduate Science and Engineering Research, 3: 5–15.
Wright SL, Thompson RC, and Galloway TS. 2013. The physical impacts of microplastics on marine organisms: a review. Environmental Pollution, 178: 483–492.
Wright SL, Ulke J, Font A, Chan KLA, and Kelly FJ. 2020. Atmospheric microplastic deposition in an urban environment and an evaluation of transport. Environment International, 136: 105411.
Yonkos LT, Friedel EA, Perez-Reyes AC, Ghosal S, and Arthur CD. 2014. Microplastics in four Estuarine Rivers in the Chesapeake Bay, U.S.A. Environmental Science & Technology, 48(24): 14195–14202.
Zobkov M, and Esiukova E. 2017. Microplastics in Baltic bottom sediments: quantification procedures and first results. Marine Pollution Bulletin, 114(2): 724–732.
Supplementary materials
Supplementary Material 1 (DOCX / 576 KB)
- Download
- 576.62 KB
Supplementary Material 2 (XLSX / 13.1 KB)
- Download
- 13.16 KB
Information & Authors
Information
Published In

FACETS
Volume 6 • Number 1 • January 2021
Pages: 26 - 43
Editor: Anne Salomon
History
Received: 24 July 2020
Accepted: 25 September 2020
Version of record online: 14 January 2021
Copyright
© 2021 Bujaczek et al. This work is licensed under a Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Data Availability Statement
All relevant data are within the paper and in the Supplementary Material.
Key Words
Sections
Subjects
Authors
Author Contributions
TB, DL, and MSR conceived and designed the study.
TB, SK, and MSR performed the experiments/collected the data.
TB, SK, and MSR analyzed and interpreted the data.
DL and MSR contributed resources.
TB, DL, and MSR drafted or revised the manuscript.
Competing Interests
The authors have declared that no competing interests exist.
Metrics & Citations
Metrics
Other Metrics
Citations
Cite As
Taylor Bujaczek, Sheldon Kolter, David Locky, and Matthew S. Ross. 2021. Characterization of microplastics and anthropogenic fibers in surface waters of the North Saskatchewan River, Alberta, Canada. FACETS.
6(): 26-43. https://doi.org/10.1139/facets-2020-0057
Export Citations
If you have the appropriate software installed, you can download article citation data to the citation manager of your choice. Simply select your manager software from the list below and click Download.
Cited by
1. High throughput application of ASTM D8332: Detailed prototype design and operating conditions for microplastic sampling of riverine systems
2. Determining ice-jam stage frequency distributions of an ungauged river reach using dendrogeomorphological data
3. Distribution and Fate of Polyethylene Microplastics Released by a Portable Toilet Manufacturer into a Freshwater Wetland and Lake
4. Sampling and analyzing microplastics in rivers: What methods are being used after a decade of research?
5. Impact of flooding on microplastic abundance and distribution in freshwater environment: a review
6. Challenge in increasing the use of animal-origin textile fibers to reduce microplastic pollution on earth
7. Microplastics in water from the confluence of tropical rivers: Overall review and a case study in Paraiba do Sul River basin
8. Levels and composition of microplastics and microfibers in the South Saskatchewan River and stormwater retention ponds in the City of Saskatoon, Canada
9. Microplastics in subsurface water and zooplankton from eight lakes in British Columbia
10. Preservation, storage, and sample preparation methods for freshwater microplastics – a comprehensive review
11. Microplastic pollution in riverine ecosystems: threats posed on macroinvertebrates
12. Elimination of a Mixture of Microplastics Using Conventional and Detergent-Assisted Coagulation
13. Microplastic pollution in two remote rivers of Türkiye
14. Environmental fate of microplastics in an urban river: Spatial distribution and seasonal variation
15. The Complex Dynamics of Microplastic Migration through Different Aquatic Environments: Subsidies for a Better Understanding of Its Environmental Dispersion
16. A fit‐for‐purpose categorization scheme for microplastic morphologies
17. Plastic waste in sandy beaches and surface water in Thanh Hoa, Vietnam: abundance, characterization, and sources
18. Estimated discharge of microplastics via urban stormwater during individual rain events
19. Evaluating community science sampling for microplastics in shore sediments of large river watersheds
20. Microplastic pollution in typical seasonal rivers in northern China: temporal variation and risk assessment
21. Microplastics in urban waters and its effects on microbial communities: a critical review
22. Change in microplastic concentration during various temporal events downstream of a combined sewage overflow and in an urban stormwater creek
23. Spatial and temporal variations of microplastic concentrations in Portland's freshwater ecosystems
24. The Occurrence of Microplastics and the Formation of Biofilms by Pathogenic and Opportunistic Bacteria as Threats in Aquaculture
25. Spatiotemporal dynamics of microplastics in an urban river network area
26. Microplastic-associated pathogens and antimicrobial resistance in environment
27. Plastic pollution in waterways and in the oceans
28. Microplastics in freshwater: A global review of factors affecting spatial and temporal variations
29. Effect of Physical Characteristics and Hydrodynamic Conditions on Transport and Deposition of Microplastics in Riverine Ecosystem
30. Impacts of Plastic Pollution on Ecosystem Services, Sustainable Development Goals, and Need to Focus on Circular Economy and Policy Interventions
31. Abundance, interaction, ingestion, ecological concerns, and mitigation policies of microplastic pollution in riverine ecosystem: A review
