Neuroprotective effect of Panax ginseng extract against cerebral ischemia–reperfusion-injury-induced oxidative stress in middle cerebral artery occlusion models
Abstract
Introduction
Materials and methods
Panax ginseng extract
PGE dose
Animals
Middle cerebral artery occlusion (MCAO) surgery
Sham surgery
Experimental protocol
Blood and tissue sampling
Tissue preparation for histopathology
Tissue homogenization
Biochemical assays
Tissue antioxidants
Catalase assay
Superoxide dismutase assay
Glutathione assay
Statistical analysis
Results
Body weight changes
| Parameters | Control (n = 9) | Sham (n = 9) | MCAO (n = 9) | MCAO + PGE treated (n = 9) |
|---|---|---|---|---|
| Body Weight (g) | 222.778 ± 5.873 | 199 ± 7.356* | 189.75 ± 4.761***/NS | 191.625 ± 4.366***/NS/NS |
| TC (mg/dL) | 132.576 ± 7.738 | 127.245 ± 7.358NS | 128.215 ± 7.190NS/NS | 121.797 ± 4.054NS/NS/NS |
| TG (mg/dL) | 65.725 ± 7.356 | 62.778 ± 6.934NS | 60.871 ± 1.986NS/NS | 64.329 ± 1.866NS/NS/NS |
| HDL (mg/dL) | 51.358 ± 2.626 | 52.785 ± 3.602NS | 42.638 ± 1.240*/* | 44.928 ± 1.815NS/NS/NS |
| LDL (mg/dL) | 68.072 ± 5.353 | 61.903 ± 8.549NS | 73.012 ± 7.666NS/NS | 64.002 ± 4.111NS/NS/NS |
| VLDL (mg/dL) | 13.145 ± 1.471 | 12.555 ± 1.386NS | 12.563 ± 0.415NS/NS | 12.865 ± 0.373NS/NS/NS |
| ALT (IU/L) | 20.137 ± 3.01 | 23.978 ± 0.678NS | 25.135 ± 0.988NS/NS | 23.779 ± 1.694NS/NS/NS |
| AST (IU/L) | 34.171 ± 1.945 | 36.333 ± 2.752NS | 40.105 ± 2.015NS/NS | 30.16 ± 3.246NS/NS/* |
| ALP (IU/L) | 42.72 ± 3.174 | 43.434 ± 2.491NS | 51.466 ± 3.174NS/NS | 41.535 ± 2.24NS/NS/* |
| Urea (mg/dL) | 33.233 ± 0.718 | 35.233 ± 0.864NS | 49.02 ± 3.19***/** | 36.671 ± 3.693NS/NS/* |
| Uric Acid (mg/dL) | 2.537 ± 0.553 | 2.739 ± 0.882NS | 4.13 ± 0.981NS/NS | 3.796 ± 1.921NS/NS/NS |
| Creatinine (mg/dL) | 0.315 ± 0.033 | 0.34 ± 0.041NS | 0.552 ± 0.047***/** | 0.401 ± 0.054NS/NSNS |
| BUN (mg/dL) | 15.529 ± 0.335 | 16.464 ± 0.404NS | 22.906 ± 1.49***/** | 15.216 ± 1.51NS/NS/*** |
| LDH (U/L) | 682.295 ± 16.106 | 696.213 ± 15.523NS | 711.078 ± 26.244NS/NS | 681.163 ± 26.904NS/NS/NS |
| CRP (mg/dL) | 9.575 ± 0.169 | 10.107 ± 0.416NS | 10.914 ± 0.735NS/NS | 10.251 ± 0.259NS/NS/NS |
| Glucose (mg/dL) | 113.743 ± 5.266 | 110.836 ± 8.648NS | 112.97 ± 13.76NS/NS | 105.881 ± 7.049NS/NS/NS |
Note: Values are presented as mean ± SEM. Significant difference between experimental groups by t test are: *p < 0.05, **p < 0.01, ***p < 0.005. NS, non-significant, compared with control/compared with sham/compared with MCAO group. MCAO, middle cerebral artery occlusion; PGE, Panax ginseng extract; TC, total cholesterol; TG, triglyceride; HDL, high density lipoprotein; LDL, low density lipoprotein; VLDL, very low density lipoprotein; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; BUN, blood urea nitrogen; LDH, lactate dehydrogenase; CRP, C-reactive protein.
Effect of PGE treatment on biochemical parameters
Effect of PGE on lipid profile
Effect of PGE on liver enzymes
Effect of PGE on kidney parameters
Effect of PGE on LDH, CRP, and glucose
Effect of PGE treatment on tissues (brain, liver, and kidney) weight, total protein, and antioxidants (catalase, superoxide dismutase, and glutathione)
Effect of PGE on brain antioxidant levels
| Parameters | Control (n = 9) | Sham (n = 9) | MCAO (n = 9) | MCAO + PGE treated (n = 9) |
|---|---|---|---|---|
| Organ weight (g) | 1.378 ± 0.077 | 1.543 ± 0.041NS | 1.661 ± 0.023*/NS | 1.703 ± 0.049**/*/NS |
| Total protein (g/dL) | 4.788 ± 0.228 | 4.762 ± 0.122NS | 4.829 ± 0.046NS/NS | 4.448 ± 0.114NS/NS/* |
| Tissue catalase (μmol/g of tissue) | 19.564 ± 1.387 | 19.135 ± 0.959NS | 18.686 ± 0.802NS/NS | 20.303 ± 0.425NS/NS/NS |
| Tissue SOD (U/g of tissue) | 65.995 ± 2.626 | 63.924 ± 2.107NS | 58.842 ± 1.984NS/NS | 61.553 ± 0.376NS/NS/NS |
| Tissue glutathione (U/g of tissue) | 0.061 ± 0.002 | 0.05766 ± 0.001NS | 0.055 ± 0.002NS/NS | 0.0611 ± 0.002NS/NS/NS |
Note: Values are presented as mean ± SEM. Significant difference between experimental groups by t test are: *p < 0.05; **p < 0.01; *** p < 0.005. NS, non-significant, compared with control/compared with sham/compared with MCAO group. MCAO, middle cerebral artery occlusion; PGE, Panax ginseng extract; SOD, superoxide dismutase.
Effect of PGE on liver antioxidant levels
| Parameters | Control (n = 9) | Sham (n = 9) | MCAO (n = 9) | MCAO + PGE treated (n = 9) |
|---|---|---|---|---|
| Left kidney weight (g) | 0.457 ± 0.054 | 0.638 ± 0.061NS | 0.669 ± 0.081NS/NS | 0.656 ± 0.019*/NS/NS |
| Right kidney weight (g) | 0.480 ± 0.062 | 0.653 ± 0.06NS | 0.676 ± 0.082NS/NS | 0.675 ± 0.017NS/NS/NS |
| Total protein (g/dL) | 4.826 ± 0.092 | 4.624 ± 0.134NS | 3.996 ± 0.178**/* | 4.5 ± 0.13NS/NS/NS |
| Tissue catalase (μmol/g of tissue) | 23.161 ± 1.394 | 22.872 ± 1.002NS | 20.406 ± 0.508NS/NS | 24.616 ± 0.587NS/NS/*** |
| Tissue SOD (U/g of tissue) | 64.924 ± 3.078 | 64.08 ± 2.858NS | 60.77 ± 1.245NS/NS | 65.402 ± 1.103NS/NS/* |
| Tissue glutathione (U/g of tissue) | 0.072 ± 0.006 | 0.0816 ± 0.007NS | 0.05 ± 0.002*/** | 0.086 ± 0.001NS/NS/*** |
Note: Values are presented as mean ± SEM. Significant difference between experimental groups by t test are: *p < 0.05; **p < 0.01; ***p < 0.005. NS, non-significant, compared with control/compared with sham/compared with MCAO group. MCAO, middle cerebral artery occlusion; PGE, Panax ginseng extract; SOD, superoxide dismutase.
Effect of PGE on kidney antioxidant levels
| Parameters | Control (n = 9) | Sham (n = 9) | MCAO (n = 9) | MCAO + PGE treated (n = 9) |
|---|---|---|---|---|
| Organ weight (g) | 4.8 ± 0.19 | 4.976 ± 0.28NS | 4.705 ± 0.138NS/NS | 5.083 ± 0.366NS/NS/NS |
| Total protein (g/dL) | 5.073 ± 0.165 | 5.27 ± 0.184NS | 4.283 ± 0.244*/* | 4.823 ± 0.481**/***/NS |
| Tissue catalase (μmol/g of tissue) | 3.766 ± 0.078 | 3.835 ± 0.232NS | 3.098 ± 0.189*/* | 4.676 ± 0.075***/*/*** |
| Tissue SOD (U/g of tissue) | 11.943 ± 0.379 | 13.021 ± 0.236* | 11.125 ± 0.251NS/*** | 13.866 ± 0.176***/*/*** |
| Tissue glutathione (U/g of tissue) | 6.866 ± 0.341 | 6.558 ± 0.222NS | 5.072 ± 0.437**/* | 7.94 ± 0.28*/*/*** |
Note: Values are presented as mean ± SEM. Significant difference between experimental groups by t test are: *p < 0.05; **p < 0.01; ***p < 0.005. NS, non-significant, compared with control/compared with sham/compared with MCAO group. MCAO, middle cerebral artery occlusion; PGE, Panax ginseng extract; SOD, superoxide dismutase.
Effect of PGE treatment on tissue (brain, liver, and kidney) histology
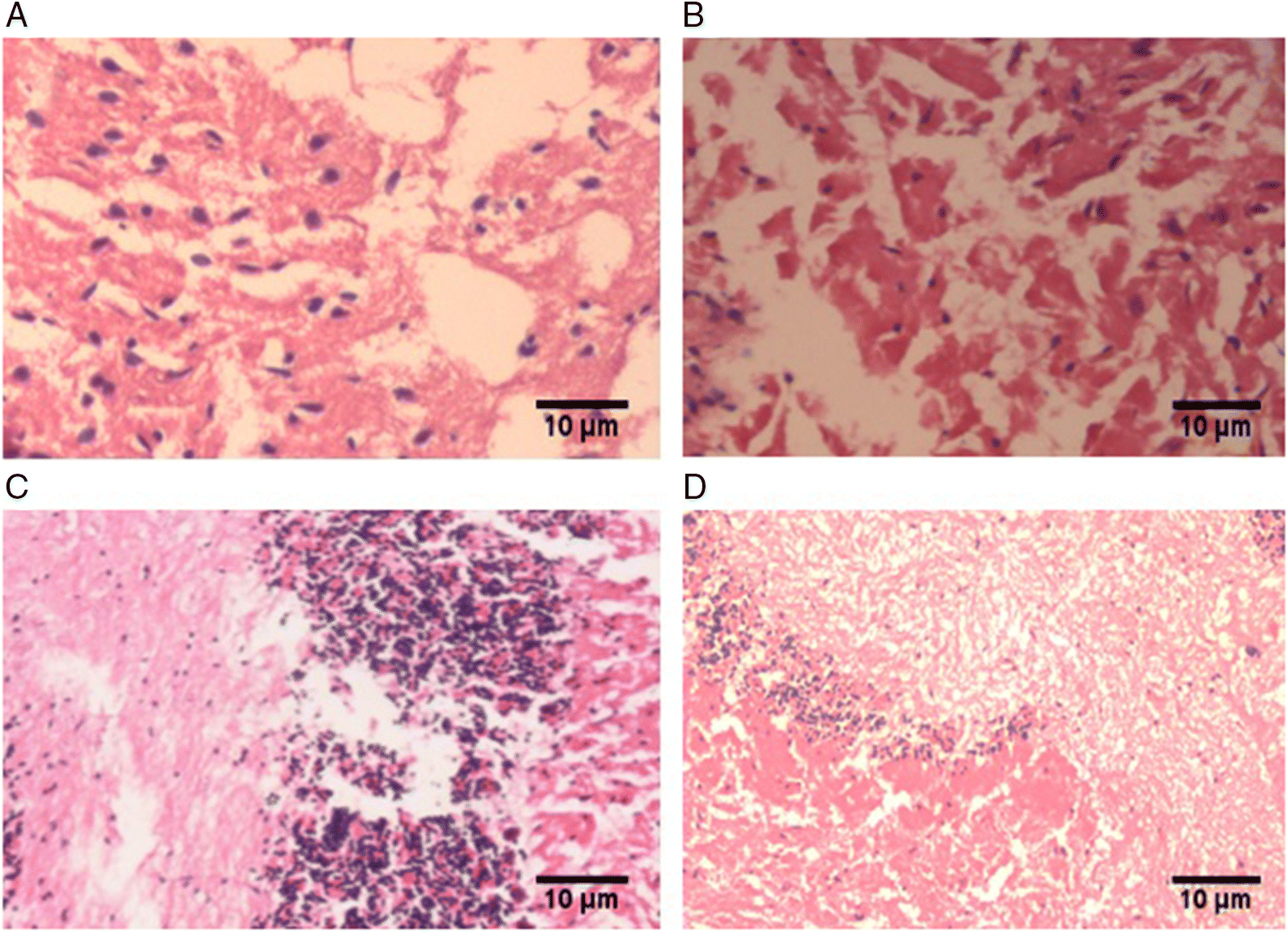
Histopathological changes in brain tissue
| Pathology type | Control (n = 9) | Sham (n = 9) | MCAO (n = 9) | MCAO + PGE treated (n = 9) |
|---|---|---|---|---|
| Cellular disintegration | — | — | +2 | — |
| Cellular swelling | — | +1 | +2 | +1 |
| Monocyte infiltration | — | +1 | +2 | +1 |
| Macrophage infiltration | — | +1 | +3 | +1 |
| PMNL degranulation | — | +1 | +3 | — |
Note: Scale: (−) absent; (+) present; (+1) minimal; (+2) mild; (+3) moderate; (+4) severe. MCAO, middle cerebral artery occlusion; PGE, Panax ginseng extract; PMNL, polymorphonuclear leucocyte.
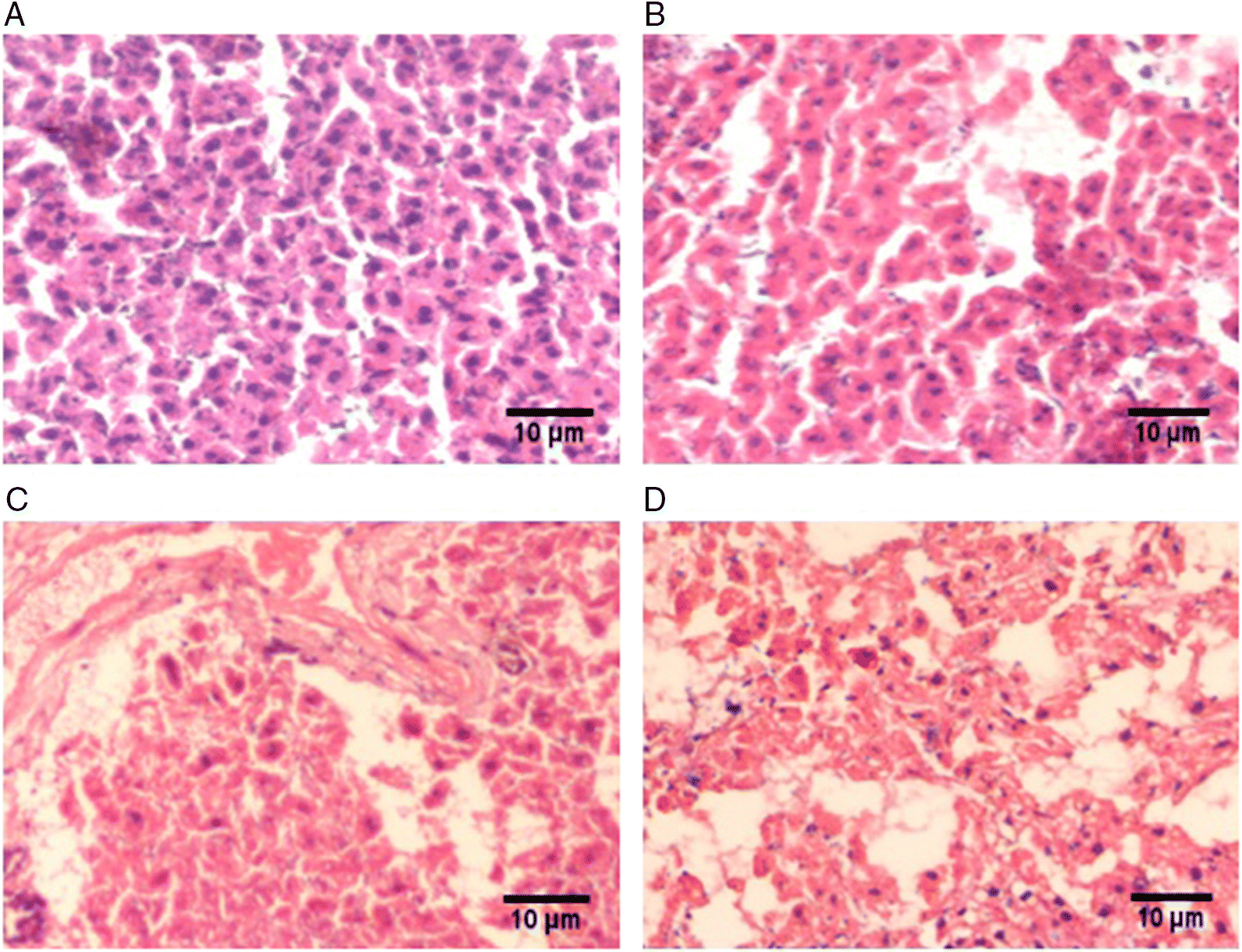
Histopathological changes in liver tissue
| Pathology type | Control (n = 9) | Sham (n = 9) | MCAO (n = 9) | MCAO + PGE treated (n = 9) |
|---|---|---|---|---|
| Cytoplasmic vacuolization | — | — | — | — |
| Nuclear condensation | — | — | — | — |
| Neutrophil infiltration | — | — | +2 | +1 |
| PMNL infiltration | — | — | +2 | +1 |
| Inflammatory changes | — | — | — | — |
Note: Scale: (+1) minimal; (+2) mild; (+3) moderate; (+4) severe. MCAO, middle cerebral artery occlusion; PGE, Panax ginseng extract; PMNL, polymorphonuclear leucocyte.
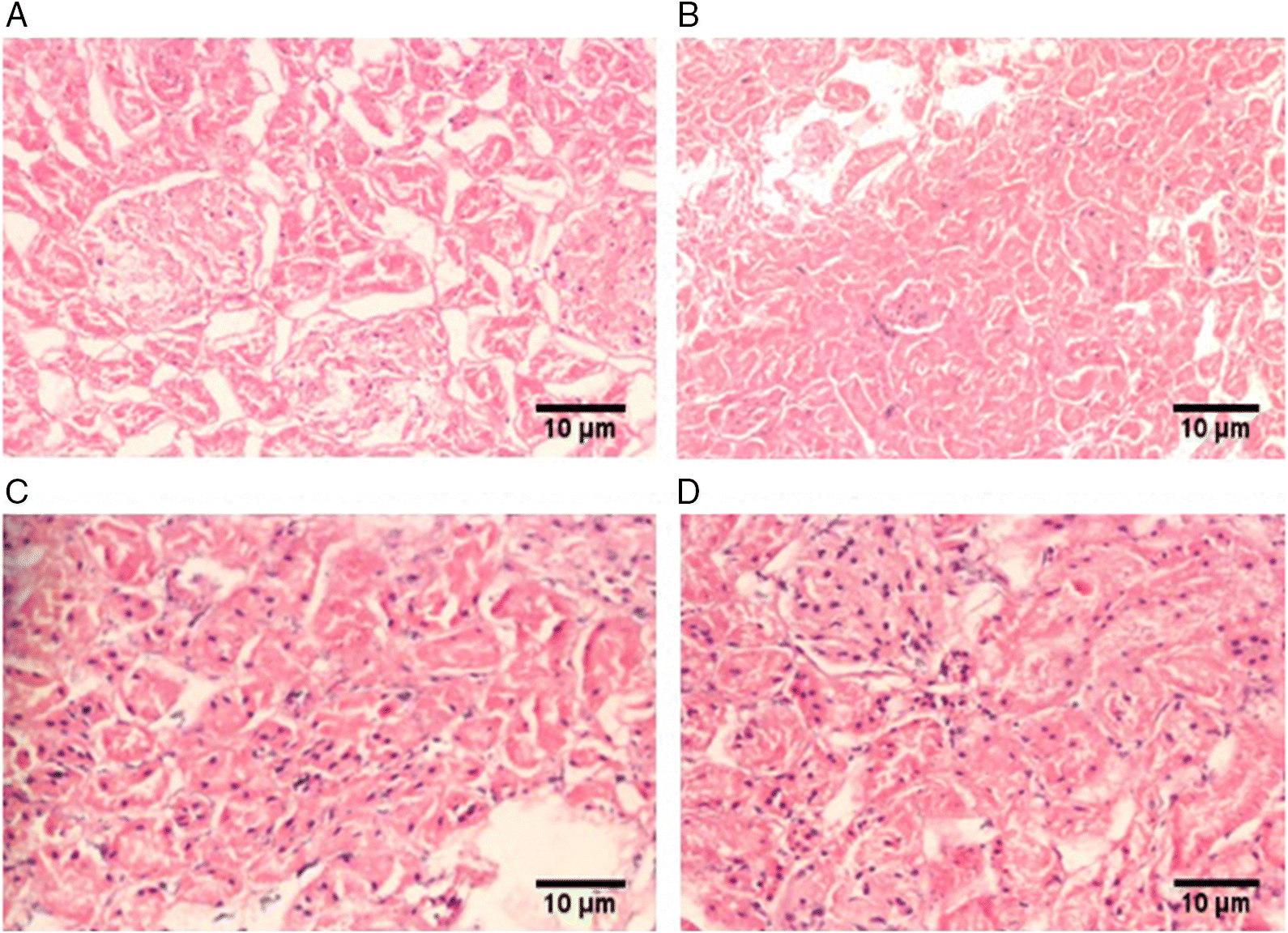
Histopathological changes in kidney tissue
| Pathology type | Control (n = 9) | Sham (n = 9) | MCAO (n = 9) | MCAO + PGE treated (n = 9) |
|---|---|---|---|---|
| Neutrophil infiltration | — | — | +2 | +1 |
| Monocyte/macrophage infiltration | — | — | +2 | +1 |
| Mesangial proliferation | — | — | — | — |
| Interstitial inflammation | — | — | — | — |
Note: Scale: +1, minimal; +2, mild; +3, moderate; +4, severe. MCAO, middle cerebral artery occlusion; PGE, Panax ginseng extract.
Discussion
Conclusion
Acknowledgements
References
Information & Authors
Information
Published In

History
Copyright
Data Availability Statement
Key Words
Sections
Subjects
Authors
Author Contributions
Competing Interests
Metrics & Citations
Metrics
Other Metrics
Citations
Cite As
Export Citations
If you have the appropriate software installed, you can download article citation data to the citation manager of your choice. Simply select your manager software from the list below and click Download.
