Introduction
The laying of eggs in the nest of an individual of the same species, known as conspecific brood parasitism (CBP), has been observed in approximately 250 bird species (
Yom-Tov 2001) and in some insects (
Zink 2000), fish (
Tariel et al. 2019), and amphibians (
Brown et al., 2008). In birds, selection is expected to favour brood parasites that make use of social information to select a suitable nest for parasitism (
Pöysä et al. 2014;
Eadie and Savard 2015). For example, CBP has been linked to host characteristics that let parasites discriminate among hosts of varying quality (
Brown and Brown 1991;
1998). Females that achieve high reproductive success frequently nest early in the breeding season (e.g.,
Blums and Clark 2004) and, indeed, parasites often target the earliest initiated nests (
Clawson et al. 1979;
Sorenson 1991;
Eadie and Savard 2015). Host experience and body condition may also serve as cues for parasites (
Paasivaara et al., 2010;
Pöysä et al. 2014; but see
Waldeck et al. 2011), because older females in good condition (e.g., with sufficient stored nutrients) may be less likely to abandon their nests than inexperienced hosts in poor condition (
Blums et al. 1997).
CBP can be costly to the fitness of the host, who must care for its young and those of the parasite. Among birds that feed their young, broods enlarged through parasitism increase the demand for parental care, which can reduce survival of offspring (
Lyon et al. 2002;
Tucker et al. 2016) and possibly that of adults (
Nur 1984; but see
Brown and Brown 1998). In contrast, costs of caring for additional, newly hatched young in species with self-feeding offspring, namely waterfowl, are likely small (
Rohwer and Freeman 1989;
Dugger and Blums 2001), although there may be costs of CBP during the nesting phase. Waterfowl lay large clutches (
Jetz et al. 2008), and incubation of clutches enlarged by parasitism can be inefficient (
Caldwell and Cornwell 1975), leading to reduced egg hatchability (
Andersson and Eriksson 1982;
Semel et al. 1988;
Eadie 1989;
Sayler 1992;
Waltho and Coulson 2015). Large clutches may take longer to incubate (
Hepp et al. 1990), forcing females to expend more energy in parental care and prolong exposure to predators (
Roy Nielsen et al. 2006a). Greater investment in incubation may ultimately have consequences to annual survival of the host (
Andersson 1984), but the idea remains speculative because effects of CBP on host survival have rarely been addressed in waterfowl (but see
Rohwer and Heusmann 1991;
Milonoff and Paananen 1993).
Assessment of whether CBP affects lifetime reproductive success of the host is required to explore whether brood parasitism is an exploitative relationship with reproductive benefits to the parasite and costs to the host (
Eadie et al. 1988;
Lyon and Eadie 2008;
2017;
Eadie and Savard 2015). In addition, fitness costs of CBP are essential inputs to determine the role of kinship and relatedness in the evolution of brood parasitism (
Andersson 1984;
McRae and Burke 1996). For example, brood parasites should avoid laying eggs in the nests of kin if CBP is costly to the host (
Zink 2000), whereas they are expected to parasitize nests of relatives when costs are low (
Andersson 2001;
Jaatinen et al. 2011).
This paper explores occurrence and correlates of CBP throughout the lifetime of a host and how the addition of foreign eggs relates to components of her fitness. We focused on a population of red-breasted mergansers (
Mergus serrator) in which CBP is an important element of breeding (
Young and Titman 1988) and where strong site fidelity has allowed adult females to be followed over their breeding histories (
Craik et al. 2020). The study had three main objectives. First, we assessed how lifetime rates of CBP varied across and within hosts by (
i) examining the level of repeatability in nest state (parasitized or nonparasitized) over a host’s lifetime and (
ii) quantifying the relationship between a host’s current nest state and that during the previous year’s nesting attempt. Second, host traits, namely date of nest initiation, age, and body mass, were assessed as potential cues used by parasites for selecting a host nest. Finally, we explored whether brood parasitism was related to host hatching success and annual survival. We relied on data from a field study in which we monitored nests of 206 red-breasted mergansers, 91 of which were followed over multiple breeding seasons.
Discussion
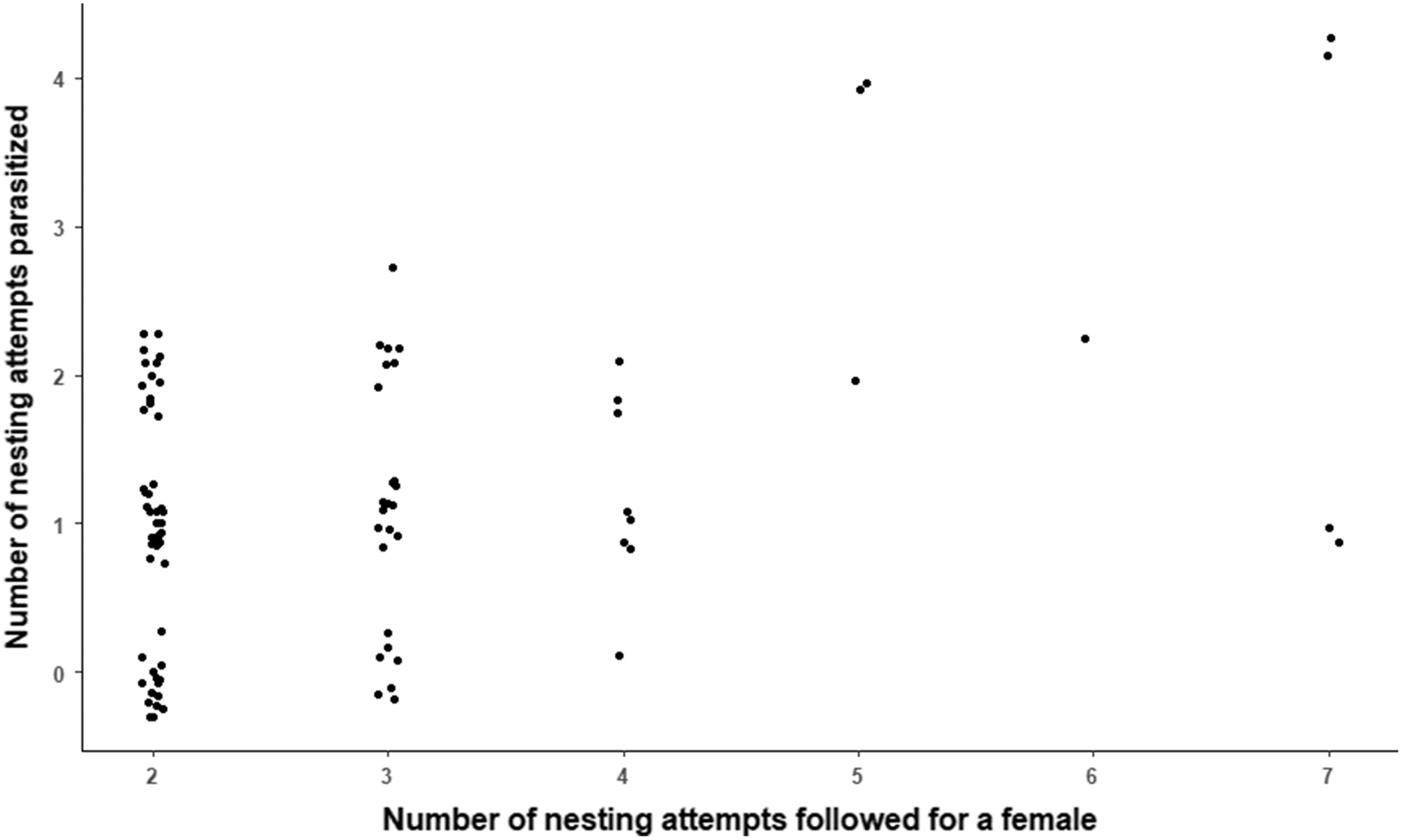
We followed nesting by a host for an average of three seasons and up to seven seasons. A couple of lines of evidence suggest that at least some females were followed for at least most of their reproductive years. First, the top-ranked mark-recapture model gave an annual survival estimate of 68%, implying that females are unlikely to live beyond 7–8 years. Indeed, only 2% of females banded on the archipelago since 2002 nested beyond their eighth year. Since female red-breasted mergansers do not begin breeding until at least their second year, the reproductive life for many hosts likely does not surpass four or five seasons. Second, capture histories for several individuals revealed an interval of time during which she was followed in most years (nesting gaps usually of ≤2 years) after which she was never observed again. Goodness-of-fit tests showed no evidence of short-term transience from the archipelago, indicating strong philopatry to the breeding site (
Anderson et al. 1992). Absence of a female during multiple years following a time interval when she nested suggests the individual had died.
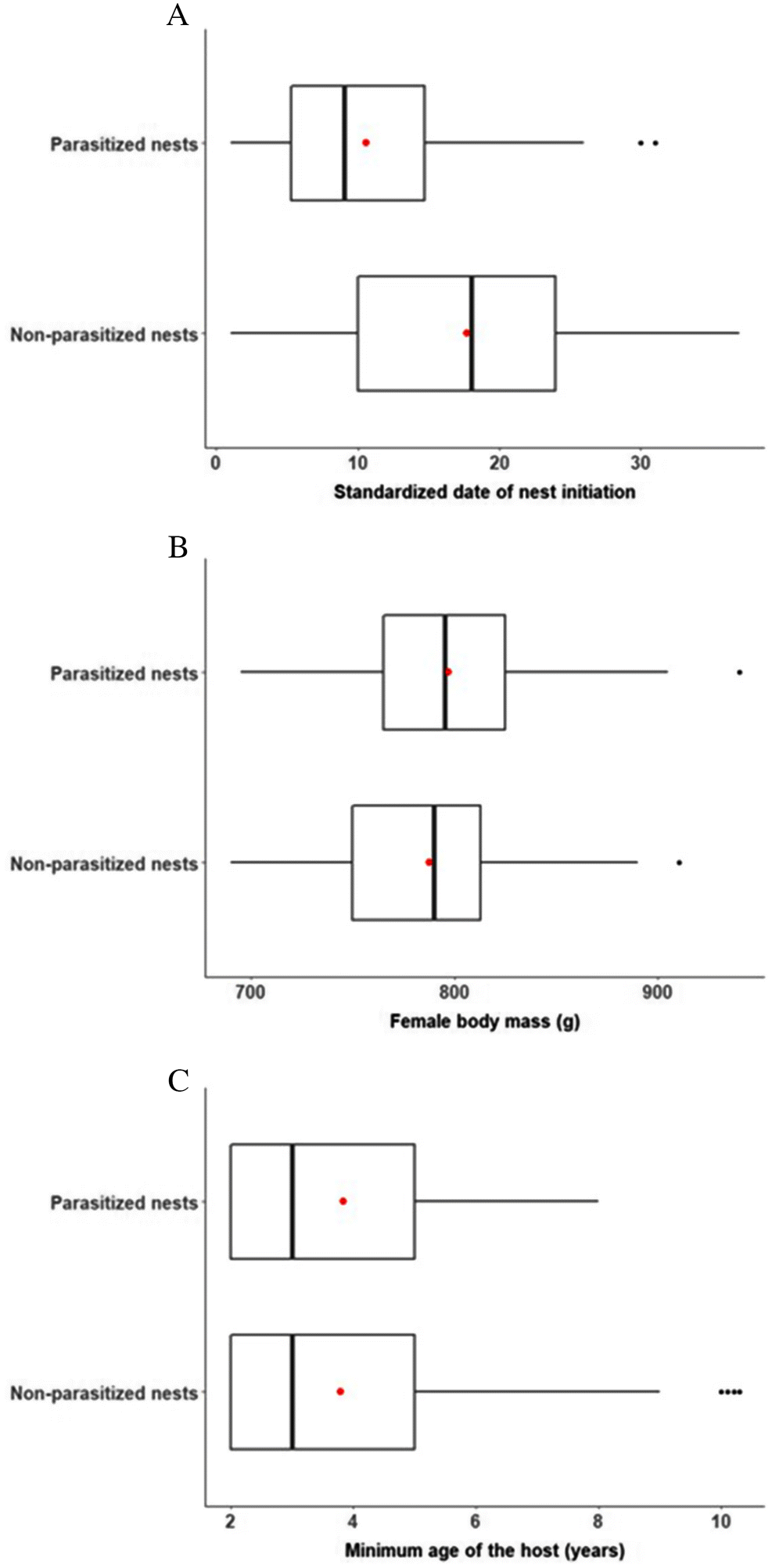
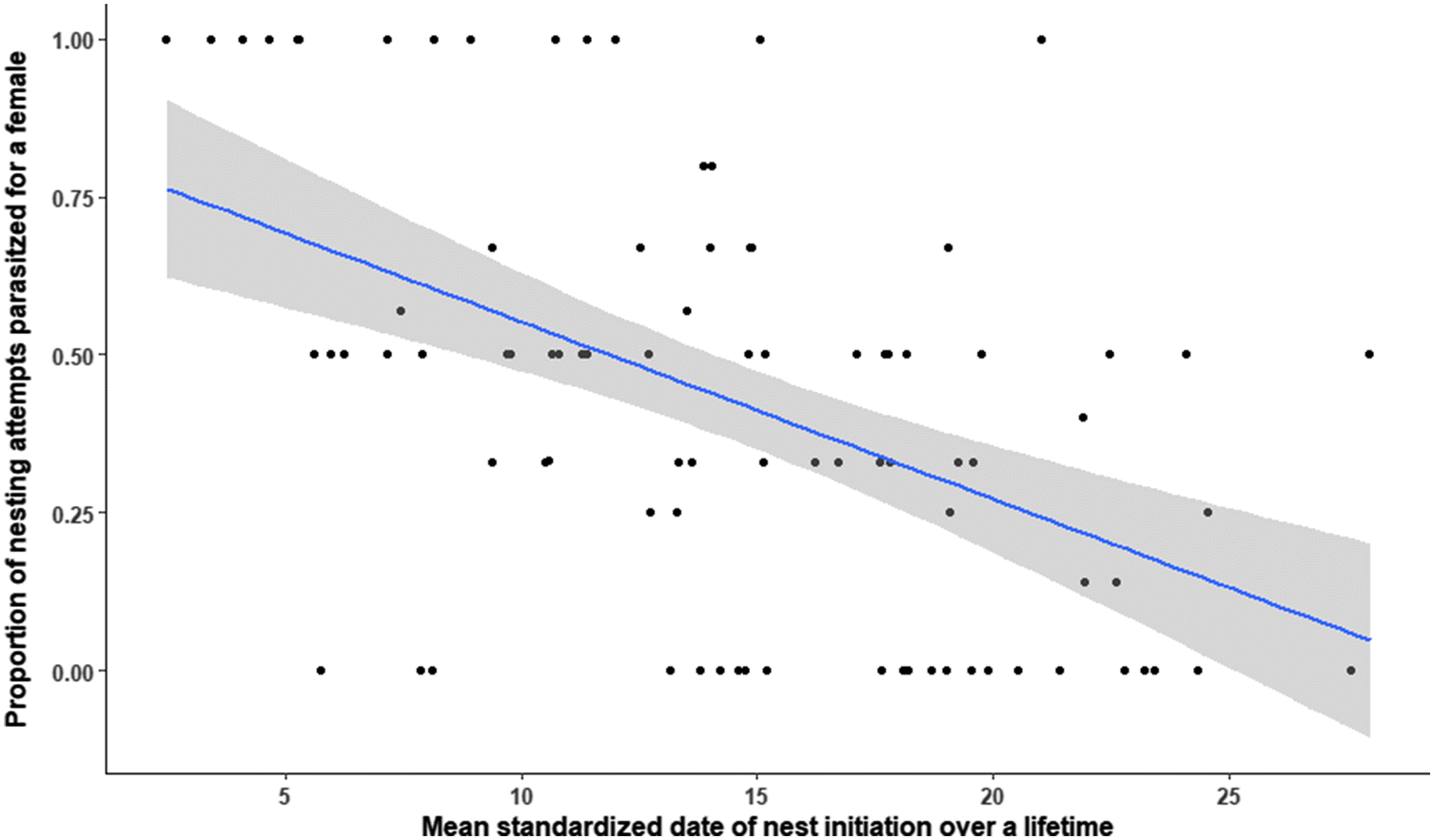
Rates of CBP over a host’s lifetime were strongly affected by dates of nest initiation because parasite activity declines throughout the season. High rates of CBP early in the season are consistent with previous reports from the study site (
Young and Titman 1988;
Thimot et al. 2020) and for a number of other waterfowl (
Clawson et al. 1979;
Sorenson 1991;
Robertson et al. 1992;
Paasivaara et al. 2010;
Eadie and Savard 2015). Some of the variability in lifetime rates of CBP reflected individual repeatability in dates of nest initiation. For example, females that typically nested early in the season had some of the highest rates of CBP at their nests, whereas hosts that were never or rarely parasitized often nested later in the season. Timing of breeding in waterfowl has been linked to host condition and experience (e.g.,
Blums and Clark 2004); however, date of nest initiation in red-breasted mergansers was unaffected by host body mass during incubation or by the host’s minimum age. An important unresolved question, then, is what drives timing of breeding in red-breasted mergansers? Body condition before breeding (
Devries et al. 2008) and timing of pair formation (
Spurr and Milne 1976) are correlates of nesting schedule in some birds, so their consideration in the red-breasted merganser may reveal important insight to factors contributing to lifetime rates of CBP for hosts in this species.
Seasonality in CBP for red-breasted mergansers may reflect adaptive behaviour by brood parasites. By parasitizing early on, females may have sufficient time to complete their own nest prior to the end of the season (
Gibbons 1986;
Lyon 1993;
Åhlund and Andersson 2001;
Reichart et al. 2010), a tactic that can favour high reproductive success relative to nesting-only or parasite-only strategies (
Sorenson 1991;
McRae 1998;
Åhlund and Andersson 2001;
Lyon and Eadie 2008). Given the strong tendency for red-breasted mergansers to parasitize nests early in the season, it may very well be that some females employ the dual tactic of parasitism plus nesting within the same season.
It seems unlikely that most brood parasites select nests of specific hosts to lay foreign eggs (i.e., host identity is not an important cue for parasites). Consistent with this idea were observations that (
i) there were no stable individual differences in occurrence of CBP across a host’s lifetime and (
ii) that a host’s nest was just as likely to change CBP states in consecutive nesting attempts as it was to remain in the same state. Host behaviour may limit opportunities for brood parasites to obtain information on the identity of the host. During egg laying (i.e., when parasite activity is greatest), red-breasted mergansers spend ∼90% of the day away from the nest and only visit it briefly to lay an egg each 1–2 days (
Noel et al. 2021), so there are likely opportunities for prospecting brood parasites to visit a nest at which the host is absent. Alternatively, brood parasites may obtain information on the identity of a nest’s host by following the host to its nest, as has been observed for some cavity-nesting waterfowl (
Heusmann et al. 1980;
Pöysä et al. 2014). Still, red-breasted mergansers parasitize experimental nests without a host (
Thimot et al. 2020) and lay in abandoned nests (
Craik and Titman 2009), confirming that host identification is not a requisite for parasitic egg laying in this bird (
Eadie 1989;
Pöysä 2003;
Odell and Eadie 2010).
There is little evidence suggesting that red-breasted mergansers try to avoid having their nest parasitized by conspecifics. Hosts can reduce the ease by which parasites find their nests by selecting sites that are isolated or less visible to conspecifics (
Roy Nielsen et al. 2006b;
Pöysä et al. 2014). However, parasitized and nonparasitized nests of red-breasted mergansers on the archipelago are surrounded by similar densities of conspecific nests (
Craik and Titman 2009) and have equally high levels of vegetative concealment (
Thimot et al. 2020). Hosts in some birds resist CBP via rejection of parasite eggs (
Emlen and Wrege 1986;
Lyon 1993;
McRae 1995;
2011;
Jamieson et al. 2000) or physically blocking a parasite’s entry to the nest (
Clawson et al. 1979;
Emlen and Wrege 1986;
McRae 1996;
Sorenson 1997;
Andersson et al. 2015). Red-breasted mergansers seem no different than other ducks in that they do not remove parasite eggs from their nest immediately after being laid (
Sayler 1992), and opportunities for hosts to resist brood parasites are limited given that hosts spend little time at the nest when parasite activity is greatest (
Noel et al. 2021). The lack of strong host defense against CBP in this species may reflect constraints due to a long egg-laying period (e.g., up to 16–17 days;
Craik et al. 2020) or lack of selective pressure given that fitness costs of CBP may be small for many hosts (see below).
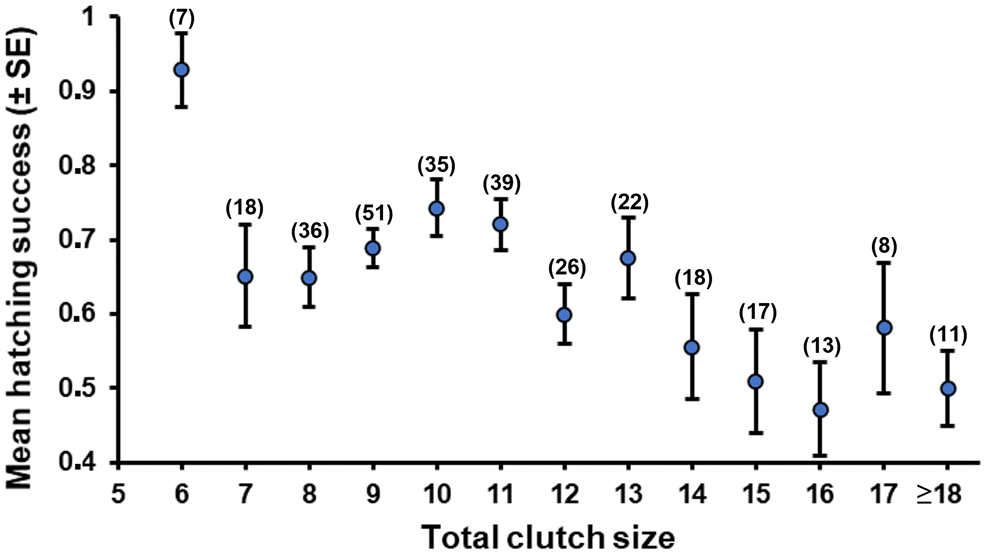
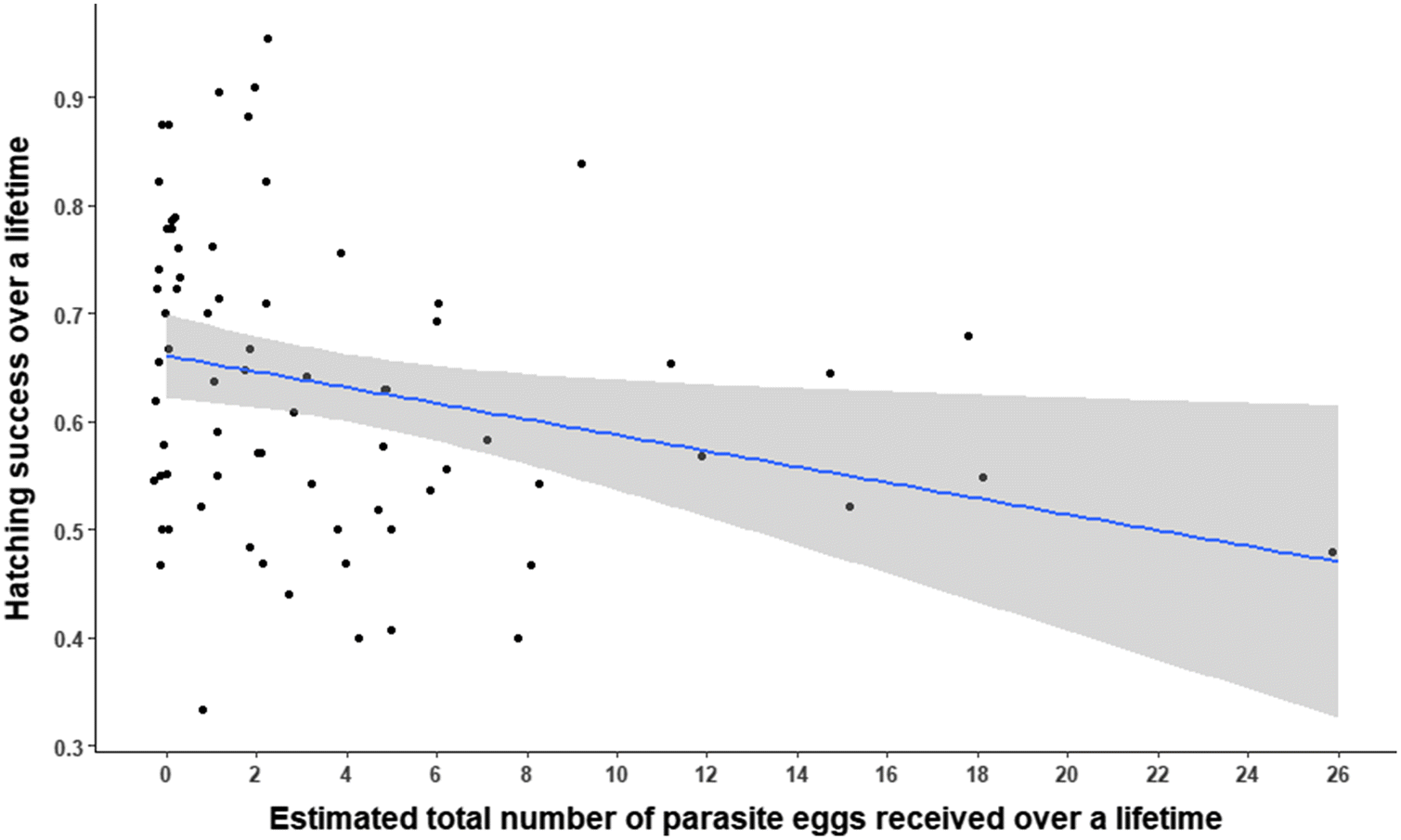
Hatching success at parasitized nests was 11% lower than at nonparasitized nests. Egg hatchability at parasitized nests on the archipelago may be lowered by (
i) embryo mortality due to inefficient incubation of large clutches (
Young and Titman 1988) or (
ii) egg displacement during incubation (
Craik et al. 2018). We caution, however, that effects of CBP on hatching success were overestimated because the clutch-size criterion missed brood parasitism in nests with ≤11 eggs, and hatching success in these nests is relatively high.
Craik et al. (2018) relied on egg-laying rates only to assess CBP from a sample of nests in this study, and they reported no difference in hatching success between parasitized and nonparasitized nests. Nonetheless, this does not take away from the fact that hatching success in the most heavily parasitized nests (≥14 eggs) that produce young was lower relative to parasitized nests with smaller clutches and nonparasitized nests. Hatching success in a nest declined with a larger number of foreign eggs, and hatching success over the lifetime of a host declined with a greater total number of parasitic eggs added to the individual’s nests. Together, these results suggest that there are measurable costs of CBP to host hatching success, though they are generally limited to the proportion of hens incubating the most heavily parasitized nests each year (20%–25% of all nests). Effects of CBP on hatching success for hens incubating smaller clutches appear to be negligible (
Craik et al. 2018).
We missed data from nests deserted during egg laying and early incubation because we relied on a sample of females identified during late incubation. Indeed, any year within an individual’s breeding history during which she was not captured may have reflected a failed breeding attempt. Nest desertion is common in this population (
Young and Titman 1986;
Craik and Titman 2009). On average, 18% of nests were abandoned during the egg-laying phase, and hens occasionally abandon nests during incubation (e.g., 18% of 106 incubated nests were deserted;
Craik and Titman 2009). A high magnitude of CBP triggers nest abandonment in some waterfowl (e.g.,
Andersson and Eriksson 1982;
Eadie 1989), and this may be the case in red-breasted mergansers.
Craik et al. (2018) found that nests receiving 2–6 experimental eggs over three days were occasionally abandoned, whereas control nests without parasitism were always incubated. Desertion of a heavily parasitized clutch may represent an important cost to fitness of a host, particularly if she does not lay the rest of her eggs elsewhere.
Annual survival probabilities for hosts tending a parasitized nest were no different than for females with a nonparasitized nest. Parents that care for large clutches may need to prolong incubation to bring eggs to term (
Hepp et al. 1990; but see
Milonoff and Paananen 1993), which extends energetic investment in parental care (
Roy Nielsen et al. 2006a) and lengthens the amount of time hosts are exposed to nest predators.
Craik et al. (2018) found no difference in incubation period between parasitized and unparasitized nests on the archipelago, while in an earlier study
Young and Titman (1986) indicated that parasitism may extend incubation by about one day in some nests, although they did not provide a statistical test. Regardless, a slightly greater incubation period is unlikely to increase risk of female predation at the nest because mammalian predators are absent from these islands and co-nesting common terns are efficient at driving avian predators away from the colony (
Young and Titman 1986).
We caution that our conclusion that broods enlarged by CBP do not affect annual survival of hosts should not extend beyond the nesting phase. The number of ducklings leaving a nest is not a reliable indicator of initial brood size for red-breasted mergansers because amalgamation of broods can occur shortly following departure from the nest (
Craik et al. 2020). Brood size may affect survival of hosts that feed their young (
Nur 1984) but may not be the case in birds wherein young are not fed (
Rohwer and Freeman 1989). Empirical data, although limited to a small number of studies with ducks (
Rohwer and Heusmann 1991) and geese (
Lessels 1986;
Lank et al. 1990;
Larsson et al. 1995) support the premise that female annual survival in waterfowl is unaffected by brood size. Unlike geese, ducks do not extend parental care beyond fledging, so an association between brood size and annual survival of brood-rearing females is unexpected in the red-breasted merganser.
Results of the multi-state mark-recapture modelling filled a significant gap in our knowledge of sea duck (Mergini) life history by providing the first estimate of apparent adult survival for the red-breasted merganser, at 0.68 per year, very close to expectations based on an average unparasitized clutch size just over 9 eggs (see fig. 10.6 in
Mallory 2015). This value places red-breasted merganser annual survival similar to that of other sea ducks like goldeneyes and buffleheads (
Bucephala spp.; 60%–67%), and in the broad but sparsely studied range of other mergansers (49%–72%), and lower than apparent survival in scoters and eiders (>75%; see review in
Mallory 2015). Principal causes of mortality for adult red-breasted mergansers are largely unknown (
Craik et al. 2020).
High-density nesting in waterfowl can lead to frequent CBP (
Sayler 1992), which in turn may lead to fitness costs for hosts (
Eadie and Savard 2015). Nest densities and rates of CBP for red-breasted mergansers on the archipelago are among the highest known for this species (
Craik et al. 2020) and are high for birds in general (
Sayler 1992;
Eadie and Savard 2015;
Lyon and Eadie 2017). Despite its common occurrence and often high magnitude at a nest, CBP had no effect on future reproduction (i.e., annual survival), and measurable costs of brood parasitism to hatching success were generally limited to the most heavily parasitized clutches, which represented ∼20%–25% nests annually. We emphasize, however, that some nest abandonment is likely triggered by heavy CBP, implying that our study may have overlooked other costs of CBP to hosts with heavily parasitized nests.
Acknowledgements
We are grateful to D. Bilodeau, A. Constantineau, M. Deveau, K. Francis, J. Haché, A. Hanks, N. Laplante, F. Leblanc, M. Mangoni, A. Rousseau, K.S. Seaborn, B. Spinney, N. Thimot, and the Titman family for their assistance in the field. Kouchibouguac National Park staff, particularly A. Beaudet, D. Gallant, the late P-É Hébert, B. Martin, and É. Tremblay provided invaluable logistical support. J.M. Eadie, B.E. Lyon, and four anonymous referees provided very helpful comments on earlier versions of the manuscript. This work was supported by the Natural Sciences and Engineering Research Council of Canada (DDG-2019-06038 to S.R.C.), New Brunswick Wildlife Trust Fund, McGill University, Bird Protection Québec, Sea Duck Joint Venture, and Université Sainte-Anne.