Introduction
The phosphonate herbicide glyphosate, which is the active ingredient in Roundup
®, is currently the most widely used herbicide in the world (
Vera et al. 2010;
Gomes et al. 2014;
Vera et al. 2014;
Gomes et al. 2016a,
2016b). In 2014, worldwide glyphosate use was 825 803 tons, 90.4% of which was used in an agricultural context (
Benbrook 2016). The development of genetically modified crops designed to make them resistant to glyphosate has increased the use of the herbicide. Glyphosate is also used in urban and household settings, exacerbating its exposure potential (
Benbrook 2016).
Glyphosate is a broad spectrum, non-selective systemic herbicide (
Vera et al. 2010;
Gomes et al. 2016b). Its principal mode of action is the inhibition of the synthesis of the 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS), an enzyme involved in the biosynthesis of the essential aromatic amino acids phenylalanine, tyrosine, and tryptophan (
Pérez et al. 2007;
Gomes et al. 2014). Because of its short half-life, glyphosate can be rapidly degraded by microbes to aminomethylphosphonic acid (AMPA), a metabolite that can interfere with the biosynthesis of chlorophyll (
Gomes et al. 2014). Glyphosate-based herbicides are typically sprayed on seedlings in the spring but may also be applied to crops prior to harvest (
Benbrook 2016). This herbicide is, thus, subject to leaching into streams and rivers, leading to pulse exposure in aquatic organisms.
In aquatic ecosystems, periphytic biofilms, or periphyton, often contain photosynthetic organisms that contribute significantly to primary production in lakes and streams (
Wetzel 1983;
Wetzel 1993;
Stevenson et al. 1996;
Battin et al. 2016). They are also biogeochemical drivers of contaminant cycling, because they include phototrophic, heterotrophic, and even chemotrophic organisms (
Desrosiers et al. 2006;
Montuelle et al. 2010;
Hamelin et al. 2011;
Lázaro et al. 2013;
Leclerc et al. 2015). These biofilms are a complex assemblage of living and dead bacteria, algae, and fungi embedded in a polysaccharide matrix that also includes organic and inorganic particles (
Wetzel 1983). These communities are attached to submerged surfaces such as plants, rocks, and sediments. Periphytic biofilms are often the first communities to be in direct contact with runoff. Cyanobacteria are ubiquitous members of freshwater biofilms, and certain members of this bacterial phylum have the ability to degrade glyphosate and use it as a source of phosphorus, which constitutes 18.2% of glyphosate’s molecular weight (
Lipok et al. 2007;
Forlani et al. 2008;
Vera et al. 2010). Biofilms are one of the most significant microbial communities at the base of food webs in shallow lakes (
Vadeboncoeur and Steinman 2002). Thus, adverse effects on upper trophic levels could occur if the structure and function of these periphytic biofilms are negatively impacted by the toxic effects of glyphosate or by its use as a source of phosphorus. Glyphosate and AMPA can, therefore, alter freshwater food webs and fish stocks by affecting periphytic biofilms when they are leached into lakes and rivers.
At early colonization stages, biofilms can be seen as open systems with low levels of nutrients and carbon internal recycling. Early organisms, both autotrophs and heterotrophs, thus, remain dependent on the availability of nutrients in the water column (
Hagerthey et al. 2011). At more mature colonization stages, microscopic ecological niches are created and nutrient exchanges with the water column decrease (
Wetzel 1993;
Hagerthey et al. 2011). In mature biofilms, communities are, therefore, more protected from external stressors (
Tlili et al. 2011;
Lozano et al. 2018). This protection can be explained by the fact that as colonization proceeds, the production of extracellular polymeric substances (EPS) by algae and bacteria, including polysaccharides, might increase, forming a barrier to the surrounding water (
Ivorra et al. 2000). However, little is known about the importance of the colonization stages of biofilms in lakes. Studies have also shown that some pesticides and metals can alter the function and composition of periphytic communities, based on the assumption that, within biofilms, different species show distinct levels of sensitivity to these contaminants (
Schmitt-Jansen and Altenburger 2005;
Tlili et al. 2010;
Lavoie et al. 2012). Even if some studies have assessed the effects of glyphosate on periphytic biofilms (
Pérez et al. 2007;
Vera et al. 2010;
Vera et al. 2014;
Lozano et al. 2018), its impact on community composition has mainly been evaluated through microscopic techniques (
Pérez et al. 2007;
Vera et al. 2010;
Lozano et al. 2018;
Smedbol et al. 2018).
The objective of this study was to identify the factors that shape the community composition of natural lake biofilms under a glyphosate pulse stress. We hypothesized that (1) mature biofilms would be less adversely impacted by glyphosate exposure because their exchanges with the water column are limited, and that (2) glyphosate exposure would shape community composition because of the varying sensitivities amongst species. To test these two hypotheses, microcosm experiments were conducted to assess the effects of glyphosate on the community composition of natural lake periphytic biofilms grown on artificial substrates at different stages of colonization (i.e., 2 months, 1 year, and 20 years). In addition to glyphosate treatments, some biofilms were also exposed to equivalent concentrations of phosphorus to compare the amount of phosphorus potentially available for algal growth in both types of exposure. Chlorophyll a content was used to assess the adverse impacts of glyphosate and community composition was determined using 18S and 16S rRNA gene analysis to target eukaryotes and cyanobacteria, respectively. To our knowledge, this was the first study to use such contrasting colonization stages, hereafter referred to as biofilm age.
Materials and methods
Study area and sampling
Biofilms were grown in situ on Teflon
® artificial substrates in Lake Croche (0.179 km
2; 45°59′N, 74°01′W), an oligotrophic Precambrian Shield lake (
Desrosiers et al. 2006;
Perron et al. 2014;
Leclerc et al. 2015). This lake was considered to be free from prior pesticide exposure, as it is geographically isolated from agricultural lands. Thus, biofilms were not exposed to glyphosate during colonization. Substrate setups (
Fig. S1) were installed as described by
Desrosiers et al. (2006) and submerged for two months (19 July 2016–11 September 2016), 1 year (20 October 2015–21 September 2016) and 20 years (1996–2016), allowing differential colonization in terms of community structure and thickness. As Teflon is an inert substrate that favours exchanges between the biofilms and their environment, the use of such artificial substrates lessens perturbations of the community, allowing it to maintain its integrity (
Desrosiers et al. 2006). Substrate setups were anchored with a clay brick at 1 m depth in the littoral zone. On the day of substrate harvesting, the colonized round mesh disks (9.6 cm
2 surface area; 70 μm pore size) were removed from the supporting structure and kept in filtered (0.45 μm) lake water. Mesh disks and their biofilms were stored in the dark at low temperature during transport to the laboratory.
Experimental design
Three distinct pulse experiments were conducted for each different biofilm age (submersion periods of 2 months, 1 year, and 20 years). Pulse experiments were chosen over chronic exposure to mimic episodic runoff events following the application of glyphosate. For each experiment, colonized mesh disks were placed in round Pyrex
® dishes (1-L capacity) filled with 600 mL of filtered (0.45 μm) lake water. Biofilms were acclimated to experimental conditions for 24 h before the addition of glyphosate or phosphorus. Biofilms were exposed to different initial concentrations of technical grade glyphosate PESTANAL
® (Sigma-Aldrich, St. Louis, Missouri, USA); 99.7% purity; CAS: 1071-83-6) or phosphorus as KH
2PO
4 (
Bregnard et al. 1996) for a period of 7 d (
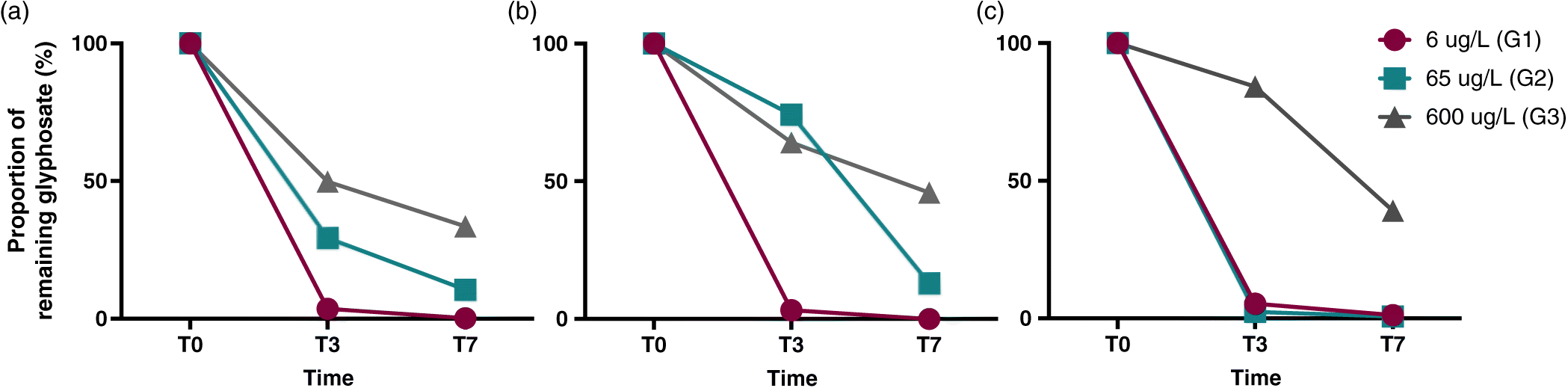
Fig. S2). Duration of exposure was chosen given preliminary results according to which only 30% of 2.5 mg/L glyphosate remained after 48 h under the same experimental conditions (
Fig. S3). We chose to directly assess the effects of the active ingredient rather than a commercial formulation, because the latter contains a mixture of additives that can alter glyphosate’s toxicity (
Vera et al. 2014). Treatments were performed in triplicate, for a total of 21 microcosms per experiment (
Fig. S2). Experimental treatments are described in
Table 1. Nominal initial concentrations of glyphosate were accurately measured in a preliminary study we conducted in 2015 under the exact same experimental conditions (
Fig. S4). In a survey of Quebec streams and rivers,
Giroux (2015) synchronized their sampling with the beginning of agricultural activities and the rainy season, as pesticides are subject to leaching to lakes and rivers following heavy rains. The highest glyphosate concentration they detected in an agricultural watershed was 6 μg/L. Because this concentration is much lower than most concentrations tested in the literature (
Pérez et al. 2007;
Vera et al. 2010;
Vera et al. 2014;
Smedbol et al. 2018), we chose it as our lowest glyphosate treatment level (
Table 1).
The microcosms were randomly distributed and incubated in a growth chamber at 23 °C, which was the temperature measured in the lake when biofilms were harvested on 31 August 2016 (average pH = 7.45 and conductivity = 9.5 μS/cm2). The day:night cycle was set to 14 h:10 h, according to illumination hours on this same day. These temperature and light parameters were maintained throughout the experiments. Constant oxygenation in the microcosms was ensured using an air pump system connected to air stones. We sampled water and periphyton from the microcosms at the very beginning of the experiment (T0), and after three (T3) and 7 d (T7).
Periphyton analysis
Two mesh disks were sampled from each microcosm on T0, T3, and T7. These two disks were brushed, pooled, and suspended in filtered (0.45 μm) lake water. Following stirring and homogenization, the suspension was subsampled and several parameters were measured.
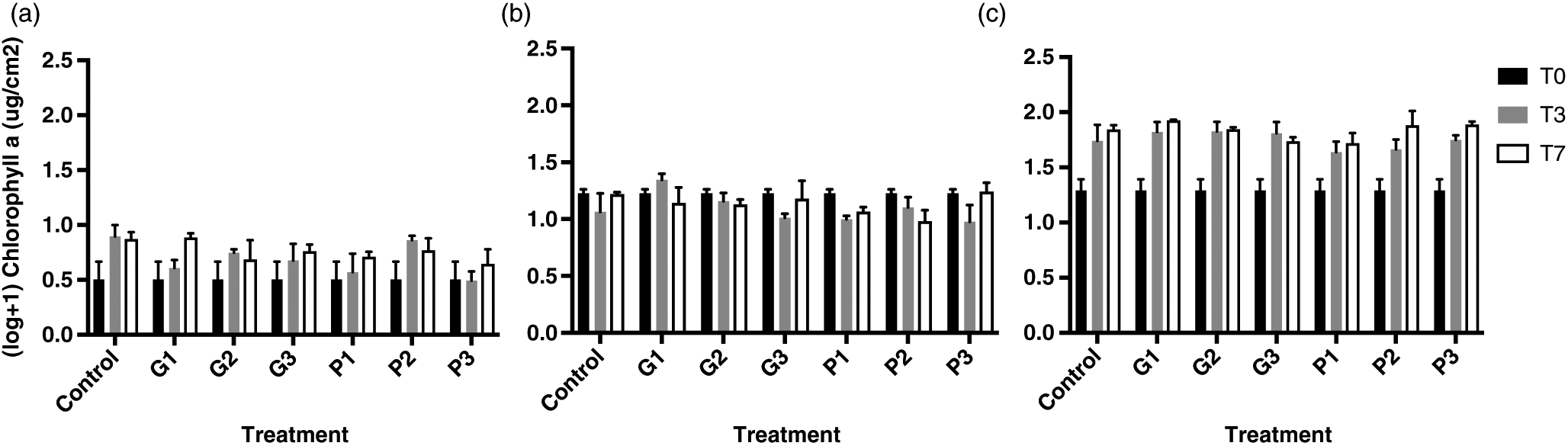
Chlorophyll a (Chla)
For Chl
a analysis, 500 μL of the periphyton suspension was filtered on GF/F glass microfibre Whatman
© filters (Whatman International Ltd., Maidstone, England) and kept frozen (−80 °C) until extraction with hot 90% ethanol (
Nusch 1980). Extracts were kept at 4 °C overnight and absorbance values were measured before and after acidification (0.01 mL HCl 1 mol/L per 1 mL of extract) at 665 nm (A
665) and 750 nm (A
750) (Spectronic Unicam UV300 UV-Visible Spectrometer (Thermo Spectronic, Rochester, New York, USA)). Surface-normalized Chl
a concentrations (μg·cm
−2) were calculated according to the following equation for each sample:
where
Ab = A
665 – A
750 before acidification,
Aa = A
665 – A
750 after acidification,
v = extraction volume (mL),
E = total sample suspension volume/filtered subsample volume (L),
L = spectrophotometer cell length (cm) and
S = mesh disk surface that was brushed (cm
2).
DNA extraction and sequence analyses
Nucleic acid extraction was performed on lyophilized biofilm samples using the DNeasy Plant Mini Kit (QIAGEN, Hilden, Germany) following the manufacturer’s instructions. Library preparation for 16S and 18S rRNA was done using Nextera XT (Illumina Inc., San Diego, California, USA), and next-generation sequencing was performed on an Illumina MiSeq (Illumina Inc., San Diego, California, USA). Reads were quality checked with FastQC (
Andrews 2010), reads were filtered with FASTX-Toolkit (
Gordon 2009) and BBMap (
Bushnell 2014), and chimeras were removed with UCHIME (
Edgar et al. 2011) in VSEARCH (
Rognes et al. 2016). Operational taxonomic unit (OTU) picking (Greengenes version 13_8, 97% identity) was performed in QIIME (
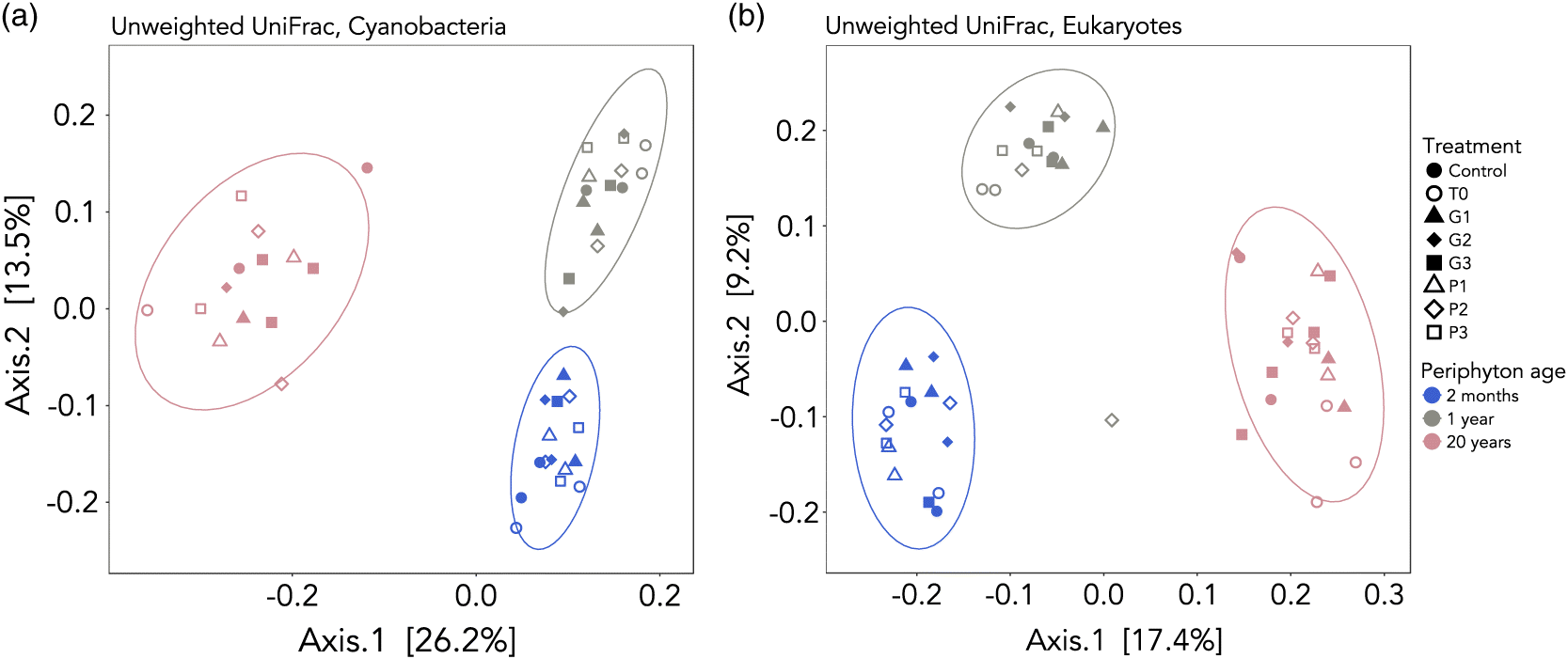
Caporaso et al. 2010). OTU tables were rarefied to 4000 and 10 000 sequences per sample for cyanobacteria-specific 16S and eukaryotic 18S, respectively. After quality filtering and rarefaction, we had 44 samples containing 916 OTUs for cyanobacteria and 49 samples containing 1920 OTUs for eukaryotes. Beta diversity analyses were performed with weighted and unweighted UniFrac (
Lozupone and Knight 2005;
Lozupone et al. 2011) as a distance metric in R (
R Core Team 2016) using the phyloseq package (
McMurdie and Holmes 2013). UniFrac uses distances between samples on a phylogenetic tree, either accounting for the relative abundance of OTUs (weighted) or not (unweighted). Community compositions among the different submersion periods were assessed. For full sequencing and read processing methods, see the
Supplementary Methods (SM.1). Sequences were submitted into the Sequence Read Archive (SRA) database (National Center for Biotechnology Information (NCBI)) (SRA accession SRP150239, BioProject PRJNA475256).
Essential aromatic amino acids
Aromatic amino acids (phenylalanine, tryptophan, and serine) concentrations were measured in 20-year-old biofilms exposed to glyphosate for 7 d using an Agilent 6430 triple quadrupole (QQQ)-LC-MS/MS (Santa Clara, California, USA) in a preliminary experiment we performed in 2015. For a detailed description of the analysis, refer to the
Supplementary Methods (SM.2).
Water analysis
Water was sampled in each microcosm for chemical analyses on T0 (immediately after glyphosate and phosphorus additions), T3, and T7. Glyphosate degradation was only determined in herbicide-treated microcosms.
Glyphosate
In 2015, we conducted the same experiment but with 20-year-old periphyton exclusively. During this preliminary study, analyses of glyphosate and AMPA were conducted by a provincial government laboratory. Glyphosate and AMPA were analyzed by liquid chromatography, post-column derivation, and fluorescence detection, with a detection limit of 0.08 μg/L for glyphosate and 0.4 μg/L for AMPA (CEAEQ method MA. 403—GlyAmp 1.0 2011-03-07) (
CEAEQ 2008). In 2016, glyphosate enzyme-linked immunosorbent assay (ELISA) test kits were purchased from Abraxis LLC (Warminster, Pennsylvania, USA) and used following the manufacturer’s instructions. The method detection limit (MDL) and the limit of quantification (LOQ) as defined by Abraxis were 0.05 μg/L and 0.15 μg/L, respectively. Derivatized standard and control solutions were used every 12 samples to ensure analytical quality. Although the former analytical method allowed us to measure accurate glyphosate concentrations, we used the latter to show loss patterns.
Phosphorus
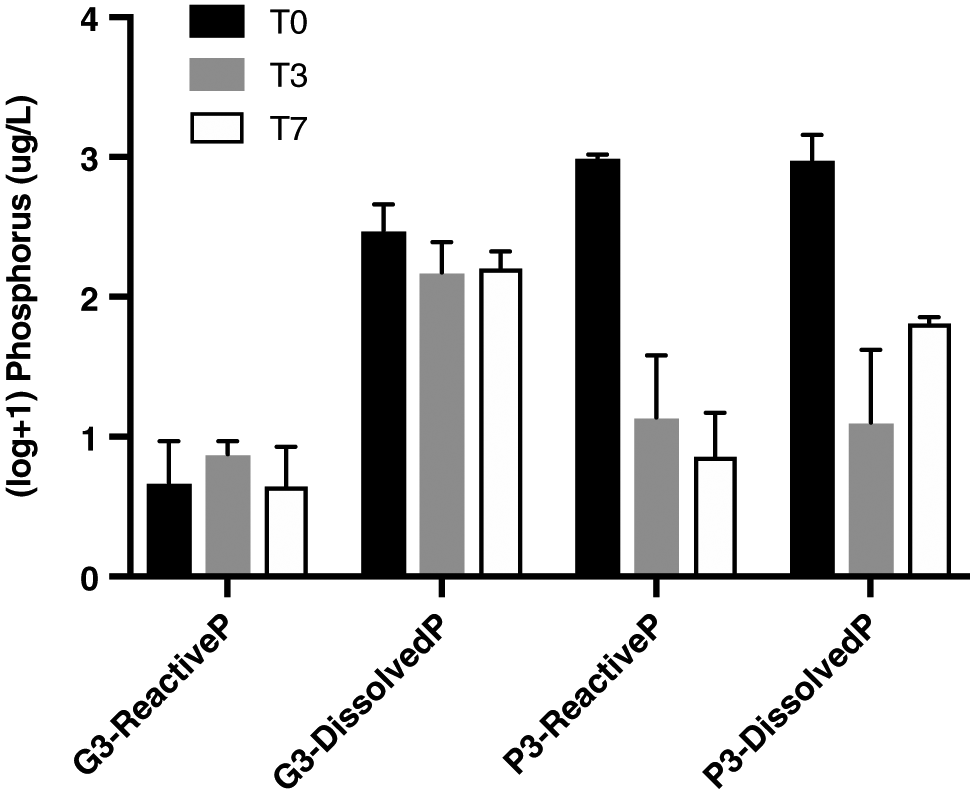
Total phosphorus (TP), dissolved phosphorus (DP), and orthophosphate levels were analyzed by flow injection analysis (Astoria2 Analyzer, Astoria-Pacific, Clackamas, Oregon, USA). Orthophosphates represent the phosphorus species that is most readily utilized by biota (
Gaffney et al. 2001). Reactive phosphorus in our experiments was, thus, measured as orthophosphates, which provide a fair estimation of the amount of phosphorus available for algal growth (
Strickland and Parsons 1972;
Carignan and Neiff 1992). We assessed (1) the amount of reactive phosphorus that was released from glyphosate degradation, and (2) whether this reactive phosphorus decreased over time as it was used by periphytic communities (deduced from decreasing concentrations over time). Water was filtered through 0.45 μm membrane filters for DP and orthophosphate analysis. TP and DP samples were analyzed after persulfate digestion in an autoclave. Orthophosphate samples were pre-acidified with 0.01 mol/L HCl (ACS grade, Fisher Scientific, Hampton, New Hampshire, USA) (
Carignan and Neiff 1992). Wash and standard solutions were used every 10 samples to ensure analytical quality.
Statistical analyses
For Chla and phosphorus results, concentrations for all treatments (Control, G1, G2, G3, P1, P2, and P3) and each sampling time (T0, T3, and T7) were compared using two-factor analysis of variance (two-way ANOVA). When required, Dunnett’s or Tukey’s post hoc tests were used for multiple comparisons. Multiple t tests were used for reactive and total dissolved phosphorus concentrations. Multiple comparisons were corrected using the Holm–Sidak method, computing adjusted p values. Prior to the analyses, normality and homoscedasticity were verified and data were log+1 transformed when these assumptions were not satisfied. Significance level was set at p < 0.05. GraphPad Prism 7 (GraphPad Software Inc., La Jolla, California, USA) was used for statistical analyses.
Principal coordinate analyses (PCoA) for 16S and 18S data from UniFrac distance matrices were calculated and visualized using the phyloseq (
McMurdie and Holmes 2013) and ggplot2 (
Wickham 2009) packages in R (
R Core Team 2016). Sample clustering hypotheses were tested using a permutational multivariate analysis of variance (permanova,
adonis()) in vegan (
Oksanen et al. 2016), as this test has been proven to be more powerful in detecting differences in community structure even when group dispersions are heterogeneous (
Anderson and Walsh 2013). Homogeneity of dispersion among sample groups was assessed using
betadisper() in vegan.
Conclusion
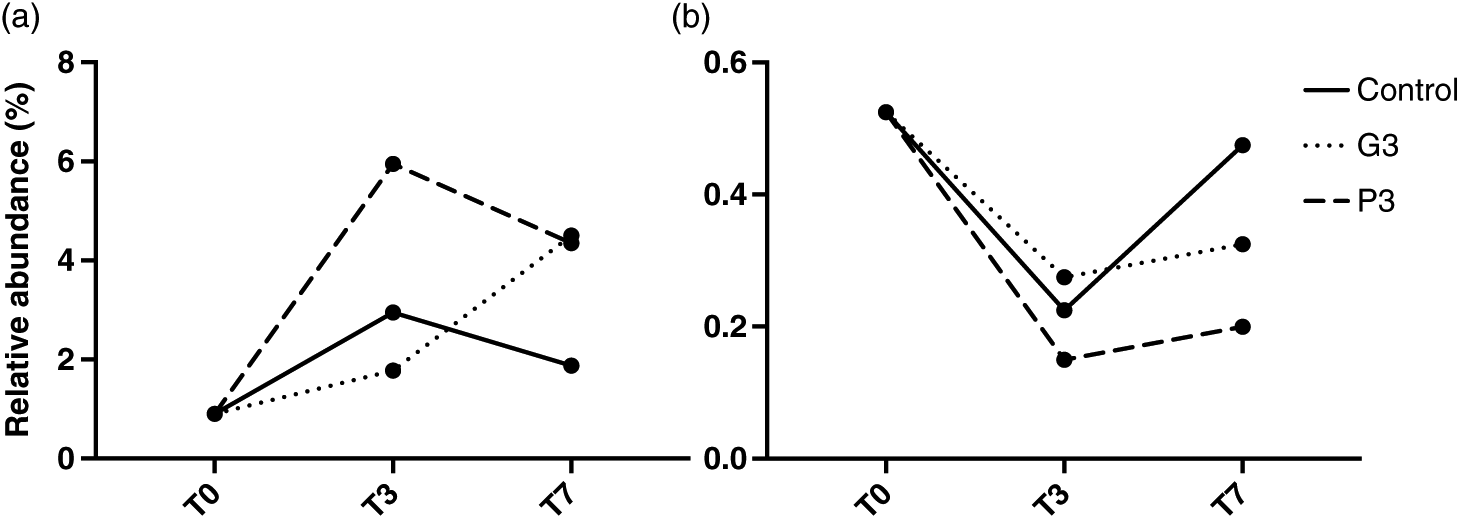
Periphytic biofilms are key organisms in ecotoxicological studies, as they are at the base of food webs, are important biogeochemical cyclers in streams and lakes, and are often the first communities to be in direct contact with runoff. This study assessed the effects of technical grade glyphosate on contrasting colonization stages of lake biofilms. The results showed that at levels that are currently detected in eastern Canadian rivers, glyphosate poses little risk for periphyton under pulse event conditions. In fact, no adverse effect was observed on Chla content. The herbicide was also found to release phosphorus in the water, but not in a bioavailable form, limiting uptake by biofilms. Periphyton age was the only factor shaping the community composition of biofilms, suggesting that the EPS layer produced by algae and bacteria was too thick to allow the penetration of glyphosate, even for the younger communities. Age and thickness likely caused important inter-experimental bias, and it is, therefore, critical to consider the colonization stage of biofilms before interpreting ecotoxicological effects. However, we identified an increase in the relative abundance of Anabaena, which has been previously found to carry a rare resistant form of the EPSPS enzyme targeted by glyphosate. This highlights the fact that some effects might be detected at a population or taxon scale, but not at a community scale. Future studies should, thus, include specific assessment of glyphosate’s toxicity using biomarkers such as reactive oxygen species or enzymes. More attention should also be given to cyanobacteria, as toxic strains seem favoured by the use of the herbicide. As specific primers allowed us to monitor the presence and abundance of cyanobacteria, future studies should focus on elucidating their mechanism of resistance. In conclusion, this study represents a step towards understanding the effects of a non-selective herbicide of rising concern on lake ecosystems.
Acknowledgements
This project was financially supported by a FRQ-NT Programme d’initiatives stratégiques pour l’innovation (ISI) grant to M.A. and D.P, by an NSERC Discovery grant to M.A, and by the Canada Research Chair program to M.A. M.K. received a scholarship through the NSERC CREATE ÉcoLac training program in Lake and Fluvial Ecology. We acknowledge the Integrated Microbiome Resource (IMR) lab at Dalhousie University (Halifax, Canada) for the library preparation of 16S and 18S amplicons, sequencing, and standard bioinformatics analysis pipeline. We acknowledge the Metabolics Core Facility at McGill University (Quebec, Canada) for the amino acid analyses. We are grateful for laboratory assistance from Dominic Bélanger, Jérémy De Bonville, Antoine Caron, Marie-Christine Lafrenière, and Serge Paquet, and field assistance from Jérémy De Bonville and Maxime Leclerc. We acknowledge the staff of the Station de biologie des Laurentides (Université de Montréal) for logistical support.